Risk prediction model for heart failure and cardiomyopathy after adjuvant trastuzumab therapy for breast cancer
- PMID: 24584736
- PMCID: PMC3959671
- DOI: 10.1161/JAHA.113.000472
Risk prediction model for heart failure and cardiomyopathy after adjuvant trastuzumab therapy for breast cancer
Abstract
Background: Adjuvant trastuzumab improves survival for women with human epidermal growth factor receptor 2-positive breast cancer, but increases risk for heart failure (HF) and cardiomyopathy (CM). However, clinical trials may underestimate HF/CM risk because they enroll younger subjects with fewer cardiac risk factors. We sought to develop a clinical risk score that identifies older women with breast cancer who are at higher risk of HF or CM after trastuzumab.
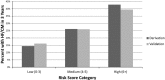
Methods and results: Using the Surveillance, Epidemiology and End Results (SEER)-Medicare database, we identified women with breast cancer who received adjuvant trastuzumab. Using a split-sample design, we used a proportional hazards model to identify candidate predictors of HF/CM in a derivation cohort. A risk score was constructed using regression coefficients, and HF/CM rates were calculated in the validation cohort. The sample consisted of 1664 older women (mean age 73.6 years) with 3-year HF/CM rate of 19.1%. A risk score consisting of age, adjuvant chemotherapy, coronary artery disease, atrial fibrillation or flutter, diabetes mellitus, hypertension, and renal failure was able to classify HF/CM risk into low (0 to 3 points), medium (4 to 5 points), and high (≥6 points) risk strata with 3-year rates of 16.2%, 26.0%, and 39.5%, respectively.
Conclusions: A 7-factor risk score was able to stratify 3-year risk of HF/CM after trastuzumab between the lowest and highest risk groups by more than 2-fold in a Medicare population. These findings will inform future research aimed at further developing a clinical risk score for HF/CM for breast cancer patients of all ages.
Keywords: breast cancer; cardiomyopathy; heart failure; trastuzumab.
Figures
Comment on
-
Heart failure and breast cancer therapies: moving towards personalized risk assessment.J Am Heart Assoc. 2014 Feb 28;3(1):e000780. doi: 10.1161/JAHA.113.000780. J Am Heart Assoc. 2014. PMID: 24584746 Free PMC article. No abstract available.
References
-
- Slamon D, Eiermann W, Robert N, Pienkowski T, Martin M, Press M, Mackey J, Glaspy J, Chan A, Pawlicki M, Pinter T, Valero V, Liu MC, Sauter G, von Minckwitz G, Visco F, Bee V, Buyse M, Bendahmane B, Tabah‐Fisch I, Lindsay MA, Riva A, Crown J. Adjuvant trastuzumab in HER2‐positive breast cancer. N Engl J Med. 2011; 365:1273-1283http://www.nejm.org/doi/suppl/10.1056/nejmoa0910383/suppl_file/nejmoa091... - DOI - PMC - PubMed
-
- Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE, Jr, Davidson NE, Tan‐Chiu E, Martino S, Paik S, Kaufman PA, Swain SM, Pisansky TM, Fehrenbacher L, Kutteh LA, Vogel VG, Visscher DW, Yothers G, Jenkins RB, Brown AM, Dakhil SR, Mamounas EP, Lingle WL, Klein PM, Ingle JN, Wolmark N. Trastuzumab plus adjuvant chemotherapy for operable HER2‐positive breast cancer. N Engl J Med. 2005; 353:1673-1684 - PubMed
-
- Piccart‐Gebhart MJ, Procter M, Leyland‐Jones B, Goldhirsch A, Untch M, Smith I, Gianni L, Baselga J, Bell R, Jackisch C, Cameron D, Dowsett M, Barrios CH, Steger G, Huang CS, Andersson M, Inbar M, Lichinitser M, Lang I, Nitz U, Iwata H, Thomssen C, Lohrisch C, Suter TM, Ruschoff J, Suto T, Greatorex V, Ward C, Straehle C, McFadden E, Dolci MS, Gelber RD. Trastuzumab after adjuvant chemotherapy in HER2‐positive breast cancer. N Engl J Med. 2005; 353:1659-1672 - PubMed
-
- Seidman A, Hudis C, Pierri MK, Shak S, Paton V, Ashby M, Murphy M, Stewart SJ, Keefe D. Cardiac dysfunction in the trastuzumab clinical trials experience. J Clin Oncol. 2002; 20:1215-1221 - PubMed
-
- Bowles E, Wellman R, Feigelson H, Onitilo A, Freedman A, Delate T, Allen L, Nekhlyudov L, Goddard K, Davis R, Habel L, Yood M, McCarty C, Magid D, Wagner E. Risk of heart failure in breast cancer patients after anthracycline and trastuzumab treatment: a retrospective cohort study. J Natl Cancer Inst. 2012; 104:1293-1305 - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous