A talin mutant that impairs talin-integrin binding in platelets decelerates αIIbβ3 activation without pathological bleeding
- PMID: 24585775
- PMCID: PMC3999757
- DOI: 10.1182/blood-2013-12-543363
A talin mutant that impairs talin-integrin binding in platelets decelerates αIIbβ3 activation without pathological bleeding
Abstract
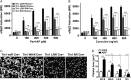
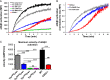
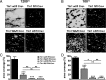
Tight regulation of integrin affinity is critical for hemostasis. A final step of integrin activation is talin binding to 2 sites within the integrin β cytoplasmic domain. Binding of talin to a membrane-distal NPxY sequence facilitates a second, weaker interaction of talin with an integrin membrane-proximal region (MPR) that is critical for integrin activation. To test the functional significance of these distinct interactions on platelet function in vivo, we generated knock-in mice expressing talin1 mutants with impaired capacity to interact with the β3 integrin MPR (L325R) or NPLY sequence (W359A). Both talin1(L325R) and talin1(W359A) mice were protected from experimental thrombosis. Talin1(L325R) mice, but not talin(W359A) mice, exhibited a severe bleeding phenotype. Activation of αIIbβ3 was completely blocked in talin1(L325R) platelets, whereas activation was reduced by approximately 50% in talin1(W359A) platelets. Quantitative biochemical measurements detected talin1(W359A) binding to β3 integrin, albeit with a 2.9-fold lower affinity than wild-type talin1. The rate of αIIbβ3 activation was slower in talin1(W359A) platelets, which consequently delayed aggregation under static conditions and reduced thrombus formation under physiological flow conditions. Together our data indicate that reduction of talin-β3 integrin binding affinity results in decelerated αIIbβ3 integrin activation and protection from arterial thrombosis without pathological bleeding.
Figures




Comment in
-
Eliminate dark side from antiplatelet therapy.Blood. 2014 Apr 24;123(17):2599-601. doi: 10.1182/blood-2014-03-560417. Blood. 2014. PMID: 24764558 No abstract available.
Similar articles
-
Talin is required for integrin-mediated platelet function in hemostasis and thrombosis.J Exp Med. 2007 Dec 24;204(13):3103-11. doi: 10.1084/jem.20071800. Epub 2007 Dec 17. J Exp Med. 2007. PMID: 18086863 Free PMC article.
-
Kindlin supports platelet integrin αIIbβ3 activation by interacting with paxillin.J Cell Sci. 2017 Nov 1;130(21):3764-3775. doi: 10.1242/jcs.205641. Epub 2017 Sep 27. J Cell Sci. 2017. PMID: 28954813 Free PMC article.
-
The antithrombotic potential of selective blockade of talin-dependent integrin alpha IIb beta 3 (platelet GPIIb-IIIa) activation.J Clin Invest. 2007 Aug;117(8):2250-9. doi: 10.1172/JCI31024. J Clin Invest. 2007. PMID: 17627302 Free PMC article.
-
Talin-dependent integrin signalling in vivo.Thromb Haemost. 2009 Jun;101(6):1020-4. Thromb Haemost. 2009. PMID: 19492142 Review.
-
Integrin activation.Biochem Soc Trans. 2008 Apr;36(Pt 2):229-34. doi: 10.1042/BST0360229. Biochem Soc Trans. 2008. PMID: 18363565 Free PMC article. Review.
Cited by
-
Gα13 Switch Region 2 Relieves Talin Autoinhibition to Activate αIIbβ3 Integrin.J Biol Chem. 2016 Dec 23;291(52):26598-26612. doi: 10.1074/jbc.M116.747279. Epub 2016 Nov 1. J Biol Chem. 2016. PMID: 27803165 Free PMC article.
-
Glatiramer acetate (copaxone) modulates platelet activation and inhibits thrombin-induced calcium influx: possible role of copaxone in targeting platelets during autoimmune neuroinflammation.PLoS One. 2014 May 2;9(5):e96256. doi: 10.1371/journal.pone.0096256. eCollection 2014. PLoS One. 2014. PMID: 24788965 Free PMC article.
-
The Rap1-RIAM-talin axis of integrin activation and blood cell function.Blood. 2016 Jul 28;128(4):479-87. doi: 10.1182/blood-2015-12-638700. Epub 2016 May 20. Blood. 2016. PMID: 27207789 Free PMC article. Review.
-
Hypofibrinogenemia with preserved hemostasis and protection from thrombosis in mice with an Fga truncation mutation.Blood. 2022 Mar 3;139(9):1374-1388. doi: 10.1182/blood.2021012537. Blood. 2022. PMID: 34905618 Free PMC article.
-
Integrin αII b tail distal of GFFKR participates in inside-out αII b β3 activation.J Thromb Haemost. 2014 Jul;12(7):1145-55. doi: 10.1111/jth.12610. Epub 2014 Jun 25. J Thromb Haemost. 2014. PMID: 24837519 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

