The wheat grain contains pectic domains exhibiting specific spatial and development-associated distribution
- PMID: 24586916
- PMCID: PMC3931795
- DOI: 10.1371/journal.pone.0089620
The wheat grain contains pectic domains exhibiting specific spatial and development-associated distribution
Abstract
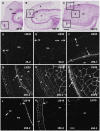
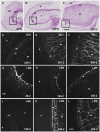
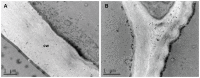
Cell walls are complex structures surrounding plant cells with a composition that varies among species and even within a species between organs, cell types and development stages. For years, cell walls in wheat grains were described as simple walls consisting mostly of arabinoxylans and mixed-linked beta glucans. Proteomic and transcriptomic studies identified enzyme families involved in the synthesis of many more cell wall polysaccharides in the wheat grains. Here we describe the discovery of pectic domains in wheat grain using monoclonal antibodies and enzymatic treatment to degrade the major cell wall polymers. Distinct spatial distributions were observed for rhamnogalacturonan I present in the endosperm and mostly in the aleurone layer and homogalacturonan especially found in the outer layers, and tight developmental regulations were unveiled. We also uncovered a massive deposition of homogalacturonan via large vesicular bodies in the seed coat (testa) beneath a thick cuticle during development. Our findings raise questions about the function of pectin in wheat grain.
Conflict of interest statement
Figures



Similar articles
-
Spatial and temporal distribution of cell wall polysaccharides during grain development of Brachypodium distachyon.Plant Sci. 2019 Mar;280:367-382. doi: 10.1016/j.plantsci.2018.12.018. Epub 2018 Dec 21. Plant Sci. 2019. PMID: 30824016
-
Comparative in situ analyses of cell wall matrix polysaccharide dynamics in developing rice and wheat grain.Planta. 2015 Mar;241(3):669-85. doi: 10.1007/s00425-014-2201-4. Epub 2014 Nov 22. Planta. 2015. PMID: 25416597 Free PMC article.
-
Change in wall composition of transfer and aleurone cells during wheat grain development.Planta. 2011 Feb;233(2):393-406. doi: 10.1007/s00425-010-1306-7. Planta. 2011. PMID: 21061017
-
Pectin: cell biology and prospects for functional analysis.Plant Mol Biol. 2001 Sep;47(1-2):9-27. Plant Mol Biol. 2001. PMID: 11554482 Review.
-
Evolution and development of cell walls in cereal grains.Front Plant Sci. 2014 Sep 11;5:456. doi: 10.3389/fpls.2014.00456. eCollection 2014. Front Plant Sci. 2014. PMID: 25309555 Free PMC article. Review.
Cited by
-
Concluding Embryogenesis After Diaspora: Seed Germination in Illicium Parviflorum.Integr Comp Biol. 2023 Dec 29;63(6):1352-1363. doi: 10.1093/icb/icad078. Integr Comp Biol. 2023. PMID: 37349968 Free PMC article.
-
Mixed-Linkage Glucan Is the Main Carbohydrate Source and Starch Is an Alternative Source during Brachypodium Grain Germination.Int J Mol Sci. 2023 Apr 6;24(7):6821. doi: 10.3390/ijms24076821. Int J Mol Sci. 2023. PMID: 37047802 Free PMC article.
-
Mutation in Brachypodium caffeic acid O-methyltransferase 6 alters stem and grain lignins and improves straw saccharification without deteriorating grain quality.J Exp Bot. 2016 Jan;67(1):227-37. doi: 10.1093/jxb/erv446. Epub 2015 Oct 3. J Exp Bot. 2016. PMID: 26433202 Free PMC article.
-
New Growth-Related Features of Wheat Grain Pericarp Revealed by Synchrotron-Based X-ray Micro-Tomography and 3D Reconstruction.Plants (Basel). 2023 Feb 24;12(5):1038. doi: 10.3390/plants12051038. Plants (Basel). 2023. PMID: 36903900 Free PMC article.
-
Evolution of the Cell Wall Gene Families of Grasses.Front Plant Sci. 2019 Oct 4;10:1205. doi: 10.3389/fpls.2019.01205. eCollection 2019. Front Plant Sci. 2019. PMID: 31681352 Free PMC article.
References
-
- Saulnier L, Guillon F, Sado P-E, Rouau X (2007) Plant cell wall polysaccharides in storage organs: xylans (food applications). In: Kamerling J, Boons G, Lee Y, Suzuki A, Taniguchi N, et al..., editors. Comprehensive glycoscience. Oxford: Elsevier. 653–689.
-
- Oury F, Carre B, Pluchard P, Berard P, Nys Y, et al. (1998) Genetic variability and stability of poultry feeding related characters in wheat, in relation to environmental variation. Agronomie 18: 139–150.
-
- Mares D, Stone B (1973a) Studies on wheat endosperm. I. Chemical composition and ultrastructure of the cell walls. Aust J Biol Sci 23: 793–812.
-
- Fincher G, Stone B (1974) A water-soluble arabinogalactan-peptide from wheat endosperm. Aust J Biol Sci 27: 117–132.
-
- Mares D, Stone B (1973b) Studies on wheat endosperm. II. Properties of the wall components and studies on their organization in the wall. Aust J Biol Sci 26: 813–830.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

