Opposite reactivity of meningeal versus cortical microvessels to the nitric oxide donor glyceryl trinitrate evaluated in vivo with two-photon imaging
- PMID: 24586970
- PMCID: PMC3938546
- DOI: 10.1371/journal.pone.0089699
Opposite reactivity of meningeal versus cortical microvessels to the nitric oxide donor glyceryl trinitrate evaluated in vivo with two-photon imaging
Abstract
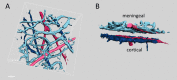
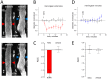
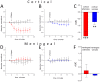
Vascular changes underlying headache in migraine patients induced by Glyceryl trinitrate (GTN) were previously studied with various imaging techniques. Despite the long history of medical and experimental use of GTN, its effects on the brain vasculature are still poorly understood presumably due to low spatial resolution of the imaging modalities used so far. We took advantage of the micrometer-scale vertical resolution of two-photon microscopy to differentiate between the vasodynamic effects of GTN on meningeal versus cortical vessels imaged simultaneously in anesthetized rats through either thinned skull or glass-sealed cranial window. Intermediate and small calibre vessels were visualized in vivo by imaging intravascular fluorescent dextran, and detection of blood flow direction allowed identification of individual arterioles and venules. We found that i.p.-injected GTN induced a transient constriction of meningeal arterioles, while their cortical counterparts were, in contrast, dilated. These opposing effects of GTN were restricted to arterioles, whereas the effects on venules were insignificant. Interestingly, the NO synthase inhibitor L-NAME did not affect the diameter of meningeal vessels but induced a constriction of cortical vessels. The different cellular environment in cortex versus meninges as well as distinct vessel wall anatomical features probably play crucial role in the observed phenomena. These findings highlight differential region- and vessel-type-specific effects of GTN on cranial vessels, and may implicate new vascular mechanisms of NO-mediated primary headaches.
Conflict of interest statement
Figures


Similar articles
-
Is Glyceryl Trinitrate, a Nitric Oxide Donor Responsible for Ameliorating the Chemical-Induced Tissue Injury In Vivo?Molecules. 2022 Jul 7;27(14):4362. doi: 10.3390/molecules27144362. Molecules. 2022. PMID: 35889233 Free PMC article.
-
Nitric oxide modulation of low-frequency oscillations in cortical vessels in FHM--a NIRS study.Headache. 2012 Jul-Aug;52(7):1146-54. doi: 10.1111/j.1526-4610.2012.02098.x. Epub 2012 Feb 21. Headache. 2012. PMID: 22352839 Free PMC article.
-
Delayed inflammation in rat meninges: implications for migraine pathophysiology.Brain. 2001 Dec;124(Pt 12):2490-502. doi: 10.1093/brain/124.12.2490. Brain. 2001. PMID: 11701602
-
Endothelial nitric oxide synthase is a site of superoxide synthesis in endothelial cells treated with glyceryl trinitrate.Br J Pharmacol. 2000 Nov;131(5):1019-23. doi: 10.1038/sj.bjp.0703665. Br J Pharmacol. 2000. PMID: 11053225 Free PMC article.
-
Investigations into the role of nitric oxide and the large intracranial arteries in migraine headache.Cephalalgia. 1997 Dec;17(8):873-95. doi: 10.1046/j.1468-2982.1997.1708873.x. Cephalalgia. 1997. PMID: 9453277 Review.
Cited by
-
Estrogen-Dependent Changes in Dura Mater Microvasculature Add New Insights to the Pathogenesis of Headache.Front Neurol. 2017 Oct 18;8:549. doi: 10.3389/fneur.2017.00549. eCollection 2017. Front Neurol. 2017. PMID: 29093699 Free PMC article. Review.
-
Harnessing Miniscope Imaging in Freely Moving Animals to Unveil Migraine Pathophysiology and Validate Novel Therapeutic Strategies.Synapse. 2024 Nov;78(6):e70001. doi: 10.1002/syn.70001. Synapse. 2024. PMID: 39567365 Free PMC article. Review.
-
Protective Effects of Hydrogen Sulfide Against the ATP-Induced Meningeal Nociception.Front Cell Neurosci. 2020 Sep 2;14:266. doi: 10.3389/fncel.2020.00266. eCollection 2020. Front Cell Neurosci. 2020. PMID: 32982692 Free PMC article.
-
Duality in response of intracranial vessels to nitroglycerin revealed in rats by imaging photoplethysmography.Sci Rep. 2023 Jul 24;13(1):11928. doi: 10.1038/s41598-023-39171-w. Sci Rep. 2023. PMID: 37488233 Free PMC article.
-
Targeted Nitric Oxide Synthase Inhibitors for Migraine.Neurotherapeutics. 2018 Apr;15(2):391-401. doi: 10.1007/s13311-018-0614-7. Neurotherapeutics. 2018. PMID: 29516436 Free PMC article. Review.
References
-
- Wolff HG (1948) Headache and other head pain. New York: Oxford University Press.
-
- Rahmann A, Wienecke T, Hansen JM, Fahrenkrug J, Olesen J, et al. (2007) Vasoactive intestinal peptide causes marked cephalic vasodilatation but does not induce migraine. Cephalalgia 28: 226–236. - PubMed
-
- Schoonman GG, van der Grong J, Kortmann C, van der Geest J, Terwindt GM, et al. (2008) Migraine headache is not associated with cerebral or meningeal vasodilatation—a 3T magnetic resonance angiography study. Brain 131: 2192–2200. - PubMed
-
- Goadsby PJ (2009) The vascular theory of migraine—a great story wrecked by the facts Brain. 132: 6–7. - PubMed
-
- Antonova M, Wienecke T, Olesen J, Ashina M (2011) Pro-inflammatory and vasoconstricting prostanoid PGF2α causes no headache in man. Cephalalgia 31: 1532–1541. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

