Neuroprotective copper bis(thiosemicarbazonato) complexes promote neurite elongation
- PMID: 24587210
- PMCID: PMC3938583
- DOI: 10.1371/journal.pone.0090070
Neuroprotective copper bis(thiosemicarbazonato) complexes promote neurite elongation
Abstract
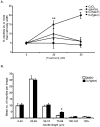
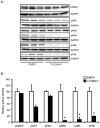
Abnormal biometal homeostasis is a central feature of many neurodegenerative disorders including Alzheimer's disease (AD), Parkinson's disease (PD), and motor neuron disease. Recent studies have shown that metal complexing compounds behaving as ionophores such as clioquinol and PBT2 have robust therapeutic activity in animal models of neurodegenerative disease; however, the mechanism of neuroprotective action remains unclear. These neuroprotective or neurogenerative processes may be related to the delivery or redistribution of biometals, such as copper and zinc, by metal ionophores. To investigate this further, we examined the effect of the bis(thiosemicarbazonato)-copper complex, Cu(II)(gtsm) on neuritogenesis and neurite elongation (neurogenerative outcomes) in PC12 neuronal-related cultures. We found that Cu(II)(gtsm) induced robust neurite elongation in PC12 cells when delivered at concentrations of 25 or 50 nM overnight. Analogous effects were observed with an alternative copper bis(thiosemicarbazonato) complex, Cu(II)(atsm), but at a higher concentration. Induction of neurite elongation by Cu(II)(gtsm) was restricted to neurites within the length range of 75-99 µm with a 2.3-fold increase in numbers of neurites in this length range with 50 nM Cu(II)(gtsm) treatment. The mechanism of neurogenerative action was investigated and revealed that Cu(II)(gtsm) inhibited cellular phosphatase activity. Treatment of cultures with 5 nM FK506 (calcineurin phosphatase inhibitor) resulted in analogous elongation of neurites compared to 50 nM Cu(II)(gtsm), suggesting a potential link between Cu(II)(gtsm)-mediated phosphatase inhibition and neurogenerative outcomes.
Conflict of interest statement
Figures






References
-
- Barnham KJ, Bush AI (2008) Metals in Alzheimer's and Parkinson's diseases. Curr Opin Chem Biol 12: 222–228. - PubMed
-
- Tamano H, Takeda A (2011) Dynamic action of neurometals at the synapse. Metallomics 3: 656–661. - PubMed
-
- Kessler H, Pajonk FG, Supprian T, Falkai P, Multhaup G, et al. (2005) [The role of copper in the pathophysiology of Alzheimer's disease]. Nervenarzt 76: 581–585. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

