Intracellular Acid-extruding regulators and the effect of lipopolysaccharide in cultured human renal artery smooth muscle cells
- PMID: 24587308
- PMCID: PMC3931831
- DOI: 10.1371/journal.pone.0090273
Intracellular Acid-extruding regulators and the effect of lipopolysaccharide in cultured human renal artery smooth muscle cells
Abstract
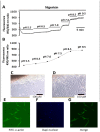
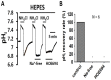
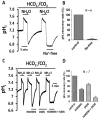
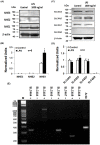
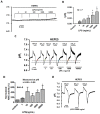
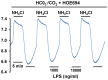
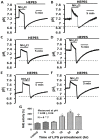
Homeostasis of the intracellular pH (pHi) in mammalian cells plays a pivotal role in maintaining cell function. Thus far, the housekeeping Na(+)-H(+) exchanger (NHE) and the Na(+)-HCO3(-) co-transporter (NBC) have been confirmed in many mammalian cells as major acid extruders. However, the role of acid-extruding regulators in human renal artery smooth muscle cells (HRASMCs) remains unclear. It has been demonstrated that lipopolysaccharide (LPS)-induced vascular occlusion is associated with the apoptosis, activating calpain and increased [Ca(2+)]i that are related to NHE1 activity in endothelia cells. This study determines the acid-extruding mechanisms and the effect of LPS on the resting pHi and active acid extruders in cultured HRASMCs. The mechanism of pHi recovery from intracellular acidosis (induced by NH4Cl-prepulse) is determined using BCECF-fluorescence in cultured HRASMCs. It is seen that (a) the resting pHi is 7.19 ± 0.03 and 7.10 ± 0.02 for HEPES- and CO2/HCO3(-)- buffered solution, respectively; (b) apart from the housekeeping NHE1, another Na(+)-coupled HCO3(-) transporter i.e. NBC, functionally co-exists to achieve acid-equivalent extrusion; (c) three different isoforms of NBC: NBCn1 (SLC4A7; electroneutral), NBCe1 (SLC4A4; electrogenic) and NBCe2 (SLC4A5), are detected in protein/mRNA level; and (d) pHi and NHE protein expression/activity are significantly increased by LPS, in both a dose- and time- dependent manner, but NBCs protein expression is not. In conclusion, it is demonstrated, for the first time, that four pHi acid-extruding regulators: NHE1, NBCn1, NBCe1 and NBCe2, co-exist in cultured HRASMCs. LPS also increases cellular growth, pHi and NHE in a dose- and time-dependent manner.
Conflict of interest statement
Figures

Similar articles
-
Functional characterization of intracellular pH regulators responsible for acid extrusion in human radial artery smooth muscle cells.Chin J Physiol. 2014 Oct 31;57(5):238-48. doi: 10.4077/CJP.2014.BAD269. Chin J Physiol. 2014. PMID: 25241983
-
Functional and molecular characterization of transmembrane intracellular pH regulators in human dental pulp stem cells.Arch Oral Biol. 2018 Jun;90:19-26. doi: 10.1016/j.archoralbio.2018.02.018. Epub 2018 Mar 6. Arch Oral Biol. 2018. PMID: 29524788
-
Functional effects of urotensin-II on intracellular pH regulators in human radial artery smooth muscle cells.Peptides. 2020 Apr;126:170236. doi: 10.1016/j.peptides.2019.170236. Epub 2019 Dec 23. Peptides. 2020. PMID: 31874233
-
Cation-coupled bicarbonate transporters.Compr Physiol. 2014 Oct;4(4):1605-37. doi: 10.1002/cphy.c130005. Compr Physiol. 2014. PMID: 25428855 Free PMC article. Review.
-
Physiology, pharmacology and pathophysiology of the pH regulatory transport proteins NHE1 and NBCn1: similarities, differences, and implications for cancer therapy.Curr Pharm Des. 2012;18(10):1345-71. doi: 10.2174/138161212799504830. Curr Pharm Des. 2012. PMID: 22360557 Review.
Cited by
-
Effects of Andrographolide on Intracellular pH Regulation, Cellular Migration, and Apoptosis in Human Cervical Cancer Cells †.Cancers (Basel). 2020 Feb 7;12(2):387. doi: 10.3390/cancers12020387. Cancers (Basel). 2020. PMID: 32046125 Free PMC article.
-
Functional and molecular mechanism of intracellular pH regulation in human inducible pluripotent stem cells.World J Stem Cells. 2018 Dec 26;10(12):196-211. doi: 10.4252/wjsc.v10.i12.196. World J Stem Cells. 2018. PMID: 30613313 Free PMC article.
-
Peroxisome Proliferator-Activated Receptor α Protects Renal Tubular Cells from Gentamicin-Induced Apoptosis via Upregulating Na+/H+ Exchanger NHE1.Mol Med. 2016 Mar;21(1):886-889. doi: 10.2119/molmed.2015.00196. Epub 2015 Nov 23. Mol Med. 2016. PMID: 26623927 Free PMC article.
References
-
- Jacobson HR (1988) Ischemic renal disease: an overlooked clinical entity? Kidney Int 34: 729–743. - PubMed
-
- Zoccali C, Mallamaci F, Finocchiaro P (2002) Atherosclerotic renal artery stenosis: epidemiology, cardiovascular outcomes, and clinical prediction rules. J Am Soc Nephrol 13: S179–183. - PubMed
-
- Vashist A, Heller EN, Brown EJ Jr, Alhaddad IA (2002) Renal artery stenosis: a cardiovascular perspective. Am Heart J 143: 559–564. - PubMed
-
- Manjunath G, Tighiouart H, Ibrahim H, MacLeod B, Salem DN, et al. (2003) Level of kidney function as a risk factor for atherosclerotic cardiovascular outcomes in the community. J Am Coll Cardiol 41: 47–55. - PubMed
-
- Conlon PJ, Little MA, Pieper K, Mark DB (2001) Severity of renal vascular disease predicts mortality in patients undergoing coronary angiography. Kidney Int 60: 1490–1497. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

