Role of endoplasmic reticulum stress in the pathogenesis of nonalcoholic fatty liver disease
- PMID: 24587654
- PMCID: PMC3930975
- DOI: 10.3748/wjg.v20.i7.1768
Role of endoplasmic reticulum stress in the pathogenesis of nonalcoholic fatty liver disease
Abstract
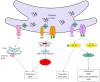
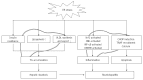
Nonalcoholic fatty liver disease (NAFLD) has emerged as a common public health problem in recent decades. However, the underlying mechanisms leading to the development of NAFLD are not fully understood. The endoplasmic reticulum (ER) stress response has recently been proposed to play a crucial role in both the development of steatosis and progression to nonalcoholic steatohepatitis. ER stress is activated to regulate protein synthesis and restore homeostatic equilibrium when the cell is stressed due to the accumulation of unfolded or misfolded proteins. However, delayed or insufficient responses to ER stress may turn physiological mechanisms into pathological consequences, including fat accumulation, insulin resistance, inflammation, and apoptosis, all of which play important roles in the pathogenesis of NAFLD. Therefore, understanding the role of ER stress in the pathogenesis of NAFLD has become a topic of intense investigation. This review highlights the recent findings linking ER stress signaling pathways to the pathogenesis of NAFLD.
Keywords: Endoplasmic reticulum stress; Nonalcoholic fatty liver disease; Nonalcoholic steatohepatitis; Unfolded protein response.
Figures
References
-
- Williams CD, Stengel J, Asike MI, Torres DM, Shaw J, Contreras M, Landt CL, Harrison SA. Prevalence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among a largely middle-aged population utilizing ultrasound and liver biopsy: a prospective study. Gastroenterology. 2011;140:124–131. - PubMed
-
- Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther. 2011;34:274–285. - PubMed
-
- Fan JG. Epidemiology of alcoholic and nonalcoholic fatty liver disease in China. J Gastroenterol Hepatol. 2013;28 Suppl 1:11–17. - PubMed
-
- Farrell GC, Wong VW, Chitturi S. NAFLD in Asia--as common and important as in the West. Nat Rev Gastroenterol Hepatol. 2013;10:307–318. - PubMed
-
- Matteoni CA, Younossi ZM, Gramlich T, Boparai N, Liu YC, McCullough AJ. Nonalcoholic fatty liver disease: a spectrum of clinical and pathological severity. Gastroenterology. 1999;116:1413–1419. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

