Drug-Induced Apoptosis: Mechanism by which Alcohol and Many Other Drugs Can Disrupt Brain Development
- PMID: 24587895
- PMCID: PMC3938204
- DOI: 10.3390/brainsci3031153
Drug-Induced Apoptosis: Mechanism by which Alcohol and Many Other Drugs Can Disrupt Brain Development
Abstract
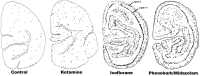
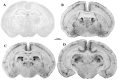
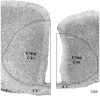
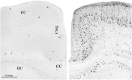
Maternal ingestion of alcohol during pregnancy can cause a disability syndrome termed Fetal Alcohol Spectrum Disorder (FASD), which may include craniofacial malformations, structural pathology in the brain, and a variety of long-term neuropsychiatric disturbances. There is compelling evidence that exposure to alcohol during early embryogenesis (4th week of gestation) can cause excessive death of cell populations that are essential for normal development of the face and brain. While this can explain craniofacial malformations and certain structural brain anomalies that sometimes accompany FASD, in many cases these features are absent, and the FASD syndrome manifests primarily as neurobehavioral disorders. It is not clear from the literature how alcohol causes these latter manifestations. In this review we will describe a growing body of evidence documenting that alcohol triggers widespread apoptotic death of neurons and oligodendroglia (OLs) in the developing brain when administered to animals, including non-human primates, during a period equivalent to the human third trimester of gestation. This cell death reaction is associated with brain changes, including overall or regional reductions in brain mass, and long-term neurobehavioral disturbances. We will also review evidence that many drugs used in pediatric and obstetric medicine, including general anesthetics (GAs) and anti-epileptics (AEDs), mimic alcohol in triggering widespread apoptotic death of neurons and OLs in the third trimester-equivalent animal brain, and that human children exposed to GAs during early infancy, or to AEDs during the third trimester of gestation, have a significantly increased incidence of FASD-like neurobehavioral disturbances. These findings provide evidence that exposure of the developing human brain to GAs in early infancy, or to alcohol or AEDs in late gestation, can cause FASD-like neurodevelopmental disability syndromes. We propose that the mechanism by which alcohol, GAs and AEDs produce neurobehavioral deficit syndromes is by triggering apoptotic death and deletion of neurons and OLs (or their precursors) from the developing brain. Therefore, there is a need for research aimed at deciphering mechanisms by which these agents trip the apoptosis trigger, the ultimate goal being to learn how to prevent these agents from causing neurodevelopmental disabilities.
Keywords: alcohol; anesthetics; anti-epileptics; apoptosis; fetal alcohol spectrum disorder.
Figures



References
-
- Ikonomidou C., Bosch F., Miksa M., Bittigau P., Vöckler J., Dikranian K., Tenkova T., Stevoska V., Turski L., Olney J.W. Blockade of NMDA receptors and apoptotic neurodegeneration in the developing brain. Science. 1999;283:70–74. - PubMed
-
- Bittigau P., Sifringer M., Genz K., Reith E., Pospischil D., Govindarajalu S., Dzietko M., Pesditschek S., Mai I., Dikranian K., et al. Antiepileptic drugs and apoptotic neurodegeneration in the developing brain. Proc. Natl. Acad. Sci. USA. 2002;99:15089–15094. doi: 10.1073/pnas.222550499. - DOI - PMC - PubMed
-
- Dikranian K., Qin Y.Q., Labruyere J., Nemmers B., Olney J.W. Ethanol-induced neuroapoptosis in the developing rodent cerebellum and related brain stem structures. Dev. Brain Res. 2005;155:1–13. - PubMed
-
- Istaphanous G.K., Howard J., Nan X., Hughes E.A., McCann J.C., McAuliffe J.J., Danzer S.C., Loepke A.W. Comparison of the neuroapoptotic properties of equipotent anesthetic concentrations of desflurane, isoflurane, or sevoflurane in neonatal mice. Anesthesiology. 2011;114:578–587. doi: 10.1097/ALN.0b013e3182084a70. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
