Apoptosis induced by islet amyloid polypeptide soluble oligomers is neutralized by diabetes-associated specific antibodies
- PMID: 24589570
- PMCID: PMC3940978
- DOI: 10.1038/srep04267
Apoptosis induced by islet amyloid polypeptide soluble oligomers is neutralized by diabetes-associated specific antibodies
Abstract
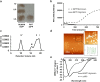
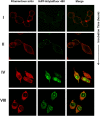
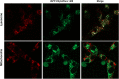
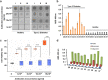
Soluble oligomeric assemblies of amyloidal proteins appear to act as major pathological agents in several degenerative disorders. Isolation and characterization of these oligomers is a pivotal step towards determination of their pathological relevance. Here we describe the isolation of Type 2 diabetes-associated islet amyloid polypeptide soluble cytotoxic oligomers; these oligomers induced apoptosis in cultured pancreatic cells, permeated model lipid vesicles and interacted with cell membranes following complete internalization. Moreover, antibodies which specifically recognized these assemblies, but not monomers or amyloid fibrils, were exclusively identified in diabetic patients and were shown to neutralize the apoptotic effect induced by these oligomers. Our findings support the notion that human IAPP peptide can form highly toxic oligomers. The presence of antibodies identified in the serum of diabetic patients confirms the pathological relevance of the oligomers. In addition, the newly identified structural epitopes may also provide new mechanistic insights and a molecular target for future therapy.
Figures

Similar articles
-
Toxic human islet amyloid polypeptide (h-IAPP) oligomers are intracellular, and vaccination to induce anti-toxic oligomer antibodies does not prevent h-IAPP-induced beta-cell apoptosis in h-IAPP transgenic mice.Diabetes. 2007 May;56(5):1324-32. doi: 10.2337/db06-1579. Epub 2007 Mar 12. Diabetes. 2007. PMID: 17353506
-
Evidence for proteotoxicity in beta cells in type 2 diabetes: toxic islet amyloid polypeptide oligomers form intracellularly in the secretory pathway.Am J Pathol. 2010 Feb;176(2):861-9. doi: 10.2353/ajpath.2010.090532. Epub 2009 Dec 30. Am J Pathol. 2010. PMID: 20042670 Free PMC article.
-
Identification of a hinge residue controlling islet amyloid polypeptide self-assembly and cytotoxicity.J Biol Chem. 2019 May 24;294(21):8452-8463. doi: 10.1074/jbc.RA118.006454. Epub 2019 Apr 11. J Biol Chem. 2019. PMID: 30975901 Free PMC article.
-
Causative factors for formation of toxic islet amyloid polypeptide oligomer in type 2 diabetes mellitus.Clin Interv Aging. 2015 Nov 19;10:1873-9. doi: 10.2147/CIA.S95297. eCollection 2015. Clin Interv Aging. 2015. PMID: 26604727 Free PMC article. Review.
-
Membrane permeabilization by Islet Amyloid Polypeptide.Chem Phys Lipids. 2009 Jul;160(1):1-10. doi: 10.1016/j.chemphyslip.2009.03.008. Epub 2009 Apr 7. Chem Phys Lipids. 2009. PMID: 19501206 Review.
Cited by
-
Protein-conformational diseases in childhood: Naturally-occurring hIAPP amyloid-oligomers and early β-cell damage in obesity and diabetes.PLoS One. 2020 Aug 24;15(8):e0237667. doi: 10.1371/journal.pone.0237667. eCollection 2020. PLoS One. 2020. PMID: 32833960 Free PMC article.
-
RAGE binds preamyloid IAPP intermediates and mediates pancreatic β cell proteotoxicity.J Clin Invest. 2018 Feb 1;128(2):682-698. doi: 10.1172/JCI85210. Epub 2018 Jan 16. J Clin Invest. 2018. PMID: 29337308 Free PMC article.
-
Role and Cytotoxicity of Amylin and Protection of Pancreatic Islet β-Cells from Amylin Cytotoxicity.Cells. 2018 Aug 6;7(8):95. doi: 10.3390/cells7080095. Cells. 2018. PMID: 30082607 Free PMC article. Review.
-
No effects without causes: the Iron Dysregulation and Dormant Microbes hypothesis for chronic, inflammatory diseases.Biol Rev Camb Philos Soc. 2018 Aug;93(3):1518-1557. doi: 10.1111/brv.12407. Epub 2018 Mar 25. Biol Rev Camb Philos Soc. 2018. PMID: 29575574 Free PMC article. Review.
-
The redox environment triggers conformational changes and aggregation of hIAPP in Type II Diabetes.Sci Rep. 2017 Mar 13;7:44041. doi: 10.1038/srep44041. Sci Rep. 2017. PMID: 28287098 Free PMC article.
References
-
- Haass C. The molecular significance of amyloid beta-peptide for Alzheimer's disease. Eur. Arch. Psychiatry Clin. Neurosci. 246, 118–123 (1996). - PubMed
-
- Conway K. A., Harper J. D. & Lansbury P. T. Accelerated in vitro fibril formation by a mutant alpha-synuclein linked to early-onset Parkinson disease. Nat. Med. 4, 1318–1320 (1998). - PubMed
-
- Chuang L. M. et al. Role of S20G mutation of amylin gene in insulin secretion, insulin sensitivity, and type 2 diabetes mellitus in Taiwanese patients. Diabetologia. 41, 1250–1251 (1998). - PubMed
-
- Terry R. D. et al. Physical basis of cognitive alterations in alzheimer's disease: Synapse loss is the major correlate of cognitive impairment. Annals of Neurology. 30, 572–580 (1991). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

