Lymphatic transport of high-density lipoproteins and chylomicrons
- PMID: 24590278
- PMCID: PMC3934183
- DOI: 10.1172/JCI71610
Lymphatic transport of high-density lipoproteins and chylomicrons
Abstract
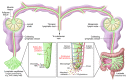
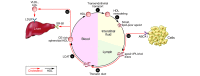
The life cycles of VLDLs and most LDLs occur within plasma. By contrast, the role of HDLs in cholesterol transport from cells requires that they readily gain access to and function within interstitial fluid. Studies of lymph derived from skin, connective tissue, and adipose tissue have demonstrated that particles as large as HDLs require transport through lymphatics to return to the bloodstream during reverse cholesterol transport. Targeting HDL for therapeutic purposes will require understanding its biology in the extravascular compartment, within the interstitium and lymph, in health and disease, and we herein review the processes that mediate the transport of HDLs and chylomicrons through the lymphatic vasculature.
Figures
References
-
- Glomset JA, Janssen ET, Kennedy R, Dobbins J. Role of plasma lecithin:cholesterol acyltransferase in the metabolism of high density lipoproteins. J Lipid Res. 1966;7(5):638–648. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

