Ex vivo expanded human regulatory T cells delay islet allograft rejection via inhibiting islet-derived monocyte chemoattractant protein-1 production in CD34+ stem cells-reconstituted NOD-scid IL2rγnull mice
- PMID: 24594640
- PMCID: PMC3940883
- DOI: 10.1371/journal.pone.0090387
Ex vivo expanded human regulatory T cells delay islet allograft rejection via inhibiting islet-derived monocyte chemoattractant protein-1 production in CD34+ stem cells-reconstituted NOD-scid IL2rγnull mice
Erratum in
- PLoS One. 2014;9(5):e98722
Abstract
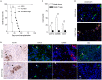
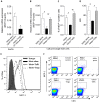
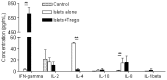
Type 1 diabetes mellitus (T1DM) is an autoimmune disease caused by immune-mediated destruction of insulin-secreting β cells of the pancreas. Near complete dependence on exogenous insulin makes T1DM very difficult to control, with the result that patients are exposed to high blood glucose and risk of diabetic complications and/or intermittent low blood glucose that can cause unconsciousness, fits and even death. Allograft transplantation of pancreatic islets restores normoglycemia with a low risk of surgical complications. However, although successful immediately after transplantation, islets are progressively lost, with most of the patients requiring exogenous insulin within 2 years post-transplant. Therefore, there is an urgent requirement for the development of new strategies to prevent islet rejection. In this study, we explored the importance of human regulatory T cells in the control of islets allograft rejection. We developed a pre-clinical model of human islet transplantation by reconstituting NOD-scid IL2rγnull mice with cord blood-derived human CD34+ stem cells and demonstrated that although the engrafted human immune system mediated the rejection of human islets, their survival was significantly prolonged following adoptive transfer of ex vivo expanded human Tregs. Mechanistically, Tregs inhibited the infiltration of innate immune cells and CD4+ T cells into the graft by down-regulating the islet graft-derived monocyte chemoattractant protein-1. Our findings might contribute to the development of clinical strategies for Treg therapy to control human islet rejection. We also show for the first time that CD34+ cells-reconstituted NOD-scid IL2rγnull mouse model could be beneficial for investigating human innate immunity in vivo.
Conflict of interest statement
Figures





References
-
- Shapiro AM, Ricordi C, Hering BJ, Auchincloss H, Lindblad R, et al. (2006) International trial of the Edmonton protocol for islet transplantation. N Engl J Med 355: 1318–1330. - PubMed
-
- Ryan EA, Paty BW, Senior PA, Bigam D, Alfadhli E, et al. (2005) Five-year follow-up after clinical islet transplantation. Diabetes 54: 2060–2069. - PubMed
-
- Fiorina P, Secchi A (2007) Pancreatic islet cell transplant for treatment of diabetes. Endocrinol Metab Clin North Am 36: 999–1013; ix. - PubMed
-
- Lamb KE, Lodhi S, Meier-Kriesche HU (2011) Long-term renal allograft survival in the United States: a critical reappraisal. Am J Transplant 11: 450–462. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

