Interaction of hyaluronan binding peptides with glycosaminoglycans in poly(ethylene glycol) hydrogels
- PMID: 24597474
- PMCID: PMC3993952
- DOI: 10.1021/bm401524h
Interaction of hyaluronan binding peptides with glycosaminoglycans in poly(ethylene glycol) hydrogels
Abstract
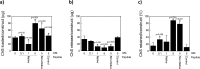
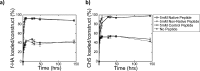
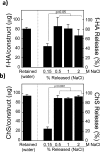
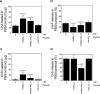
This study investigates the incorporation of hyaluronan (HA) binding peptides into poly(ethylene glycol) (PEG) hydrogels as a mechanism to bind and retain hyaluronan for applications in tissue engineering. The specificity of the peptide sequence (native RYPISRPRKRC vs non-native RPSRPRIRYKC), the role of basic amino acids, and specificity to hyaluronan over other GAGs in contributing to the peptide-hyaluronan interaction were probed through experiments and simulations. Hydrogels containing the native or non-native peptide retained hyaluronan in a dose-dependent manner. Ionic interactions were the dominating mechanism. In diH2O the peptides interacted strongly with HA and chondroitin sulfate, but in phosphate buffered saline the peptides interacted more strongly with HA. For cartilage tissue engineering, chondrocyte-laden PEG hydrogels containing increasing amounts of HA binding peptide and exogenous HA had increased retention and decreased loss of cell-secreted proteoglycans in and from the hydrogel at 28 days. This new matrix-interactive hydrogel platform holds promise for tissue regeneration.
Figures




References
-
- Leach J. B.; Schmidt C. E. In Encyclopedia of Biomaterials and Biomedical Engineering; Wnek G. E., Bowlin G. L., Eds.; Informa Healthcare USA: New York, 2008; pp 1421–1431.
-
- Chen W. Y.; Abatangelo G. Wound Repair Regen. 1999, 7, 79–89. - PubMed
-
- Lapcik L. Jr.; Lapcik L.; De Smedt S.; Demeester J.; Chabrecek P. Chem. Rev. 1998, 98, 2663–2684. - PubMed
-
- Kirker K. R.; Luo Y.; Nielson J. H.; Shelby J.; Prestwich G. D. Biomaterials 2002, 23, 3661–3671. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

