Analysis of host gene expression changes reveals distinct roles for the cytoplasmic domain of the Epstein-Barr virus receptor/CD21 in B-cell maturation, activation, and initiation of virus infection
- PMID: 24600013
- PMCID: PMC4019091
- DOI: 10.1128/JVI.03099-13
Analysis of host gene expression changes reveals distinct roles for the cytoplasmic domain of the Epstein-Barr virus receptor/CD21 in B-cell maturation, activation, and initiation of virus infection
Abstract
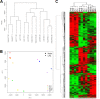
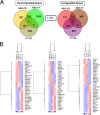
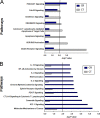
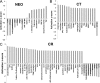
Epstein-Barr virus (EBV) attachment to human CD21 on the B-cell surface initiates infection. Whether CD21 is a simple tether or conveys vital information to the cell interior for production of host factors that promote infection of primary B cells is controversial, as the cytoplasmic fragment of CD21 is short, though highly conserved. The ubiquity of CD21 on normal B cells, the diversity of this population, and the well-known resistance of primary B cells to gene transfer technologies have all impeded resolution of this question. To uncover the role(s) of the CD21 cytoplasmic domain during infection initiation, the full-length receptor (CD21=CR), a mutant lacking the entire cytoplasmic tail (CT), and a control vector (NEO) were stably expressed in two pre-B-cell lines that lack endogenous receptor. Genome-wide transcriptional analysis demonstrated that stable CD21 surface expression alone (either CR or CT) produced multiple independent changes in gene expression, though both dramatically decreased class I melanoma-associated antigen (MAGE) family RNAs and upregulated genes associated with B-cell differentiation (e.g., C2TA, HLA-II, IL21R, MIC2, CD48, and PTPRCAP/CD45-associated protein). Temporal analysis spanning 72 h revealed that not only CR- but also CT-expressing lines initiated latency. In spite of this, the number and spectrum of transcripts altered in CR- compared with CT-bearing lines at 1 h after infection further diverged. Differential modulation of immediate early cellular transcripts (e.g., c-Jun and multiple histones), both novel and previously linked to CD21-initiated signaling, as well as distinct results from pathway analyses support a separate role for the cytoplasmic domain in initiation of intracellular signals.
Importance: Membrane proteins that mediate virus attachment tether virus particles to the cell surface, initiating infection. In addition, upon virus interaction such proteins may transmit signals to the interior of the cell that support subsequent steps in the infection process. Here we show that expression of the Epstein-Barr virus B-cell attachment receptor, CD21, in B cells that lack this receptor results in significant changes in gene expression, both before and rapidly following EBV-CD21 interaction. These changes translate into major signaling pathway alterations that are predicted to support stable infection.
Figures




References
-
- Rickinson AB, Kieff E. 2007. Epstein-Barr Virus, p 2655–2700 In Knipe DM, Howley PM. (ed), Fields virology, vol 2 Lippincott, Williams & Wilkins, Philadelphia, PA
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

