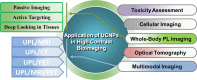
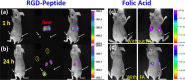
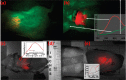
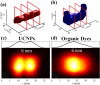
Upconversion nanoparticles: design, nanochemistry, and applications in theranostics
- PMID: 24605868
- PMCID: PMC4039352
- DOI: 10.1021/cr400425h
Upconversion nanoparticles: design, nanochemistry, and applications in theranostics
Figures
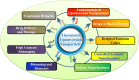
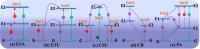
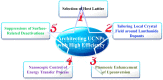
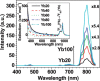













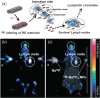
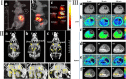


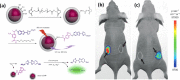


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

