Delayed formation of zero-valent selenium nanoparticles by Bacillus mycoides SeITE01 as a consequence of selenite reduction under aerobic conditions
- PMID: 24606965
- PMCID: PMC3975340
- DOI: 10.1186/1475-2859-13-35
Delayed formation of zero-valent selenium nanoparticles by Bacillus mycoides SeITE01 as a consequence of selenite reduction under aerobic conditions
Abstract
Background: Selenite (SeO32-) oxyanion shows severe toxicity to biota. Different bacterial strains exist that are capable of reducing SeO32- to non-toxic elemental selenium (Se0), with the formation of Se nanoparticles (SeNPs). These SeNPs might be exploited for technological applications due to their physico-chemical and biological characteristics. The present paper discusses the reduction of selenite to SeNPs by a strain of Bacillus sp., SeITE01, isolated from the rhizosphere of the Se-hyperaccumulator legume Astragalus bisulcatus.
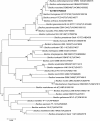
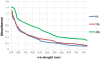
Results: Use of 16S rRNA and GyrB gene sequence analysis positioned SeITE01 phylogenetically close to B. mycoides. On agarized medium, this strain showed rhizoid growth whilst, in liquid cultures, it was capable of reducing 0.5 and 2.0 mM SeO32- within 12 and 24 hours, respectively. The resultant Se0 aggregated to form nanoparticles and the amount of Se0 measured was equivalent to the amount of selenium originally added as selenite to the growth medium. A delay of more than 24 hours was observed between the depletion of SeO32 and the detection of SeNPs. Nearly spherical-shaped SeNPs were mostly found in the extracellular environment whilst rarely in the cytoplasmic compartment. Size of SeNPs ranged from 50 to 400 nm in diameter, with dimensions greatly influenced by the incubation times. Different SeITE01 protein fractions were assayed for SeO32- reductase capability, revealing that enzymatic activity was mainly associated with the membrane fraction. Reduction of SeO32- was also detected in the supernatant of bacterial cultures upon NADH addition.
Conclusions: The selenite reducing bacterial strain SeITE01 was attributed to the species Bacillus mycoides on the basis of phenotypic and molecular traits. Under aerobic conditions, the formation of SeNPs were observed both extracellularly or intracellularly. Possible mechanisms of Se0 precipitation and SeNPs assembly are suggested. SeO32- is proposed to be enzymatically reduced to Se0 through redox reactions by proteins released from bacterial cells. Sulfhydryl groups on peptides excreted outside the cells may also react directly with selenite. Furthermore, membrane reductases and the intracellular synthesis of low molecular weight thiols such as bacillithiols may also play a role in SeO32- reduction. Formation of SeNPs seems to be the result of an Ostwald ripening mechanism.
Figures

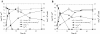



References
-
- Fordyce FM. Selenium deficiency and toxicity in the environment. Essent Med Geology. 2013;16:375–416.
-
- Ralston NVC, Ralston CR, Blackwell JL III, Raymond LJ. Dietary and tissue selenium in relation to methylmercury toxicity. Neurotoxicology. 2008;29:802.811. - PubMed
-
- Keller EA. Environmental Geology. 9. Upper Saddle River, NJ, USA: Prentice Hall; 2000.
-
- Craig PJ, Maher W. In: Organometallic Compounds in the Environment. 2. Craig PJ, editor. Chichester: Wiley; 2003. Organoselenium compounds in the environment; pp. 391–398.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

