Hormonal networks involved in apical hook development in darkness and their response to light
- PMID: 24616725
- PMCID: PMC3935338
- DOI: 10.3389/fpls.2014.00052
Hormonal networks involved in apical hook development in darkness and their response to light
Abstract
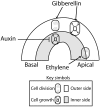
In darkness, the dicot seedlings produce an apical hook as result of differential cell division and extension at opposite sides of the hypocotyl. This hook protects the apical meristem from mechanical damage during seedling emergence from the soil. In darkness, gibberellins act via the DELLA-PIF (PHYTOCHROME INTERACTING FACTORs) pathway, and ethylene acts via the EIN3/EIL1 (ETHYLENE INSENSITIVE 3/EIN3 like 1)-HLS1 (HOOKLESS 1) pathway to control the asymmetric accumulation of auxin required for apical hook formation and maintenance. These core pathways form a network with multiple points of connection. Light perception by phytochromes and cryptochromes reduces the activity of PIFs and (COP1) CONSTITUTIVE PHOTOMORPHOGENIC 1-both required for hook formation in darkness-, lowers the levels of gibberellins, and triggers hook opening as a component of the switch between heterotrophic and photoautotrophic development. Apical hook opening is thus a suitable model to study the convergence of endogenous and exogenous signals on the control of cell division and cell growth.
Keywords: Arabidopsis; COP1; PIFs; apical hook; auxin; ethylene; gibberellins; photoreceptors.
Figures



References
-
- Abel S., Nguyen M. D., Chow W., Theologis A. (1995). Asc4, a primary indoleacetic acid-responsive gene encoding 1-aminocyclopropane-1-carboxylate synthase in Arabidopsis thaliana: structural characterization, expression in Escherichia coli, and expression characteristics in response to auxin. J. Biol. Chem. 270, 19093–19099 10.1074/jbc.270.32.19093 - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

