Chitin activates parallel immune modules that direct distinct inflammatory responses via innate lymphoid type 2 and γδ T cells
- PMID: 24631157
- PMCID: PMC4019510
- DOI: 10.1016/j.immuni.2014.02.003
Chitin activates parallel immune modules that direct distinct inflammatory responses via innate lymphoid type 2 and γδ T cells
Abstract
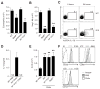
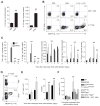
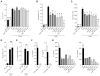
Chitin, a polysaccharide constituent of many allergens and parasites, initiates innate type 2 lung inflammation through incompletely defined pathways. We show that inhaled chitin induced expression of three epithelial cytokines, interleukin-25 (IL-25), IL-33, and thymic stromal lymphopoietin (TSLP), which nonredundantly activated resident innate lymphoid type 2 cells (ILC2s) to express IL-5 and IL-13 necessary for accumulation of eosinophils and alternatively activated macrophages (AAMs). In the absence of all three epithelial cytokines, ILC2s normally populated the lung but failed to increase IL-5 and IL-13. Although eosinophils and AAMs were attenuated, neutrophil influx remained normal without these epithelial cytokines. Genetic ablation of ILC2s, however, enhanced IL-1β, TNFα, and IL-23 expression, increased activation of IL-17A-producing γδ T cells, and prolonged neutrophil influx. Thus, chitin elicited patterns of innate cytokines that targeted distinct populations of resident lymphoid cells, revealing divergent but interacting pathways underlying the tissue accumulation of specific types of inflammatory myeloid cells.
Copyright © 2014 Elsevier Inc. All rights reserved.
Figures




Comment in
-
Innate immune cells to the help.Immunity. 2014 Mar 20;40(3):313-4. doi: 10.1016/j.immuni.2014.02.010. Immunity. 2014. PMID: 24656044
-
Innate lymphoid cells: breathing into allergic inflammation.Nat Rev Immunol. 2014 May;14(5):281. doi: 10.1038/nri3668. Epub 2014 Apr 11. Nat Rev Immunol. 2014. PMID: 24722478 No abstract available.
References
-
- Da Silva CA, Chalouni C, Williams A, Hartl D, Lee CG, Elias JA. Chitin is a size-dependent regulator of macrophage TNF and IL-10 production. J Immunol. 2009;182:3573–3582. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

