Functional integration of acute myeloid leukemia into the vascular niche
- PMID: 24637335
- PMCID: PMC4167983
- DOI: 10.1038/leu.2014.109
Functional integration of acute myeloid leukemia into the vascular niche
Abstract
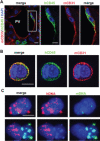
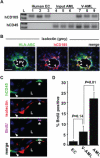
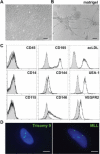
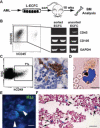
Vascular endothelial cells are a critical component of the hematopoietic microenvironment that regulates blood cell production. Recent studies suggest the existence of functional cross-talk between hematologic malignancies and vascular endothelium. Here we show that human acute myeloid leukemia (AML) localizes to the vasculature in both patients and in a xenograft model. A significant number of vascular tissue-associated AML cells (V-AML) integrate into vasculature in vivo and can fuse with endothelial cells. V-AML cells acquire several endothelial cell-like characteristics, including the upregulation of CD105, a receptor associated with activated endothelium. Remarkably, endothelial-integrated V-AML shows an almost fourfold reduction in proliferative activity compared with non-vascular-associated AML. Primary AML cells can be induced to downregulate the expression of their hematopoietic markers in vitro and differentiate into phenotypically and functionally defined endothelial-like cells. After transplantation, these leukemia-derived endothelial cells are capable of giving rise to AML. These novel functional interactions between AML cells and normal endothelium along with the reversible endothelial cell potential of AML suggest that vascular endothelium may serve as a previously unrecognized reservoir for AML.
Figures


References
-
- Burnett A, Wetzler M, Lowenberg B. Therapeutic advances in acute myeloid leukemia. J Clin Oncol. 2011 Feb 10;29(5):487–494. - PubMed
-
- Buccisano F, Maurillo L, Del Principe MI, Del Poeta G, Sconocchia G, Lo-Coco F, et al. Prognostic and therapeutic implications of minimal residual disease detection in acute myeloid leukemia. Blood. 2012 Jan 12;119(2):332–341. - PubMed
-
- Hanahan D, Coussens LM. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell. 2012 Mar 20;21(3):309–322. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

