Role of BRCA1 in brain development
- PMID: 24639535
- PMCID: PMC3977248
- DOI: 10.1073/pnas.1400783111
Role of BRCA1 in brain development
Abstract
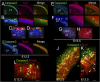
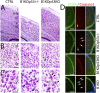
Breast cancer susceptibility gene 1 (BRCA1) is a breast and ovarian cancer tumor suppressor whose loss leads to DNA damage and defective centrosome functions. Despite its tumor suppression functions, BRCA1 is most highly expressed in the embryonic neuroepithelium when the neural progenitors are highly proliferative. To determine its functional significance, we deleted BRCA1 in the developing brain using a neural progenitor-specific driver. The phenotype is characterized by severe agenesis of multiple laminated cerebral structures affecting most notably the neocortex, hippocampus, cerebellum, and olfactory bulbs. Major phenotypes are caused by excess apoptosis, as these could be significantly suppressed by the concomitant deletion of p53. Certain phenotypes attributable to centrosomal and cell polarity functions could not be rescued by p53 deletion. A double KO with the DNA damage sensor kinase ATM was able to rescue BRCA1 loss to a greater extent than p53. Our results suggest distinct apoptotic and centrosomal functions of BRCA1 in neural progenitors, with important implications to understand the sensitivity of the embryonic brain to DNA damage, as well as the developmental regulation of brain size.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Monje ML, Mizumatsu S, Fike JR, Palmer TD. Irradiation induces neural precursor-cell dysfunction. Nat Med. 2002;8(9):955–962. - PubMed
-
- Yamazaki JN, Schull WJ. Perinatal loss and neurological abnormalities among children of the atomic bomb. Nagasaki and Hiroshima revisited, 1949 to 1989. JAMA. 1990;264(5):605–609. - PubMed
-
- Li H, et al. A role for endogenous and radiation-induced DNA double-strand breaks in p53-dependent apoptosis during cortical neurogenesis. Radiat Res. 2008;169(5):513–522. - PubMed
-
- Tanori M, et al. Developmental and oncogenic radiation effects on neural stem cells and their differentiating progeny in mouse cerebellum. Stem Cells. 2013;31(11):2506–2516. - PubMed
-
- Okamoto M, et al. Effect of radiation on the development of immature hippocampal neurons in vitro. Radiat Res. 2009;172(6):718–724. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

