Pharmacology and anti-addiction effects of the novel κ opioid receptor agonist Mesyl Sal B, a potent and long-acting analogue of salvinorin A
- PMID: 24641310
- PMCID: PMC4292965
- DOI: 10.1111/bph.12692
Pharmacology and anti-addiction effects of the novel κ opioid receptor agonist Mesyl Sal B, a potent and long-acting analogue of salvinorin A
Abstract
Background and purpose: Acute activation of κ opioid (KOP) receptors results in anticocaine-like effects, but adverse effects, such as dysphoria, aversion, sedation and depression, limit their clinical development. Salvinorin A, isolated from the plant Salvia divinorum, and its semi-synthetic analogues have been shown to have potent KOP receptor agonist activity and may induce a unique response with similar anticocaine addiction effects as the classic KOP receptor agonists, but with a different side effect profile.
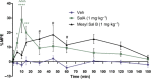
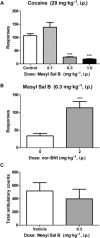
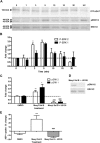
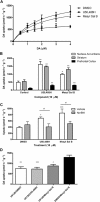
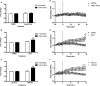
Experimental approach: We evaluated the duration of effects of Mesyl Sal B in vivo utilizing antinociception assays and screened for cocaine-prime induced cocaine-seeking behaviour in self-administering rats to predict anti-addiction effects. Cellular transporter uptake assays and in vitro voltammetry were used to assess modulation of dopamine transporter (DAT) function and to investigate transporter trafficking and kinase signalling pathways modulated by KOP receptor agonists.
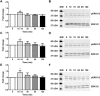
Key results: Mesyl Sal B had a longer duration of action than SalA, had anti-addiction properties and increased DAT function in vitro in a KOP receptor-dependent and Pertussis toxin-sensitive manner. These effects on DAT function required ERK1/2 activation. We identified differences between Mesyl Sal B and SalA, with Mesyl Sal B increasing the Vmax of dopamine uptake without altering cell-surface expression of DAT.
Conclusions and implications: SalA analogues, such as Mesyl Sal B, have potential for development as anticocaine agents. Further tests are warranted to elucidate the mechanisms by which the novel salvinorin-based neoclerodane diterpene KOP receptor ligands produce both anti-addiction and adverse side effects.
Linked articles: This article is part of a themed section on Opioids: New Pathways to Functional Selectivity. To view the other articles in this section visit http://dx.doi.org/10.1111/bph.2015.172.issue-2.
Keywords: Salvia divinorum; addiction; cocaine self-administration; dopamine transporter; drug seeking; nociception; salvinorin A; κ opioid receptor.
© 2014 The British Pharmacological Society.
Figures

Similar articles
-
Kappa Opioid Receptor Agonist Mesyl Sal B Attenuates Behavioral Sensitization to Cocaine with Fewer Aversive Side-Effects than Salvinorin A in Rodents.Molecules. 2018 Oct 11;23(10):2602. doi: 10.3390/molecules23102602. Molecules. 2018. PMID: 30314288 Free PMC article.
-
The 2-methoxy methyl analogue of salvinorin A attenuates cocaine-induced drug seeking and sucrose reinforcements in rats.Eur J Pharmacol. 2013 Nov 15;720(1-3):69-76. doi: 10.1016/j.ejphar.2013.10.050. Epub 2013 Nov 4. Eur J Pharmacol. 2013. PMID: 24201308 Free PMC article.
-
Salvinorin A regulates dopamine transporter function via a kappa opioid receptor and ERK1/2-dependent mechanism.Neuropharmacology. 2014 Nov;86:228-40. doi: 10.1016/j.neuropharm.2014.07.016. Epub 2014 Aug 10. Neuropharmacology. 2014. PMID: 25107591 Free PMC article.
-
Salvinorin A analogs and other κ-opioid receptor compounds as treatments for cocaine abuse.Adv Pharmacol. 2014;69:481-511. doi: 10.1016/B978-0-12-420118-7.00012-3. Adv Pharmacol. 2014. PMID: 24484985 Free PMC article. Review.
-
Salvinorin a and related compounds as therapeutic drugs for psychostimulant-related disorders.Curr Drug Abuse Rev. 2014;7(2):128-32. doi: 10.2174/1874473708666150107122741. Curr Drug Abuse Rev. 2014. PMID: 25563442 Review.
Cited by
-
Synthetic Studies of Neoclerodane Diterpenes from Salvia divinorum: Identification of a Potent and Centrally Acting μ Opioid Analgesic with Reduced Abuse Liability.J Med Chem. 2016 Dec 22;59(24):11027-11038. doi: 10.1021/acs.jmedchem.6b01235. Epub 2016 Dec 13. J Med Chem. 2016. PMID: 27958743 Free PMC article.
-
Combination of Clinically Utilized Kappa-Opioid Receptor Agonist Nalfurafine With Low-Dose Naltrexone Reduces Excessive Alcohol Drinking in Male and Female Mice.Alcohol Clin Exp Res. 2019 Jun;43(6):1077-1090. doi: 10.1111/acer.14033. Epub 2019 May 2. Alcohol Clin Exp Res. 2019. PMID: 30908671 Free PMC article.
-
The analgesic and anti-inflammatory effects of Salvinorin A analogue β-tetrahydropyran Salvinorin B in mice.Eur J Pain. 2017 Jul;21(6):1039-1050. doi: 10.1002/ejp.1002. Epub 2017 Feb 3. Eur J Pain. 2017. PMID: 28158929 Free PMC article.
-
Involvement of Activated Brain Stress Responsive Systems in Excessive and "Relapse" Alcohol Drinking in Rodent Models: Implications for Therapeutics.J Pharmacol Exp Ther. 2018 Jul;366(1):9-20. doi: 10.1124/jpet.117.245621. Epub 2018 Apr 18. J Pharmacol Exp Ther. 2018. PMID: 29669731 Free PMC article. Review.
-
Development of Diphenethylamines as Selective Kappa Opioid Receptor Ligands and Their Pharmacological Activities.Molecules. 2020 Nov 2;25(21):5092. doi: 10.3390/molecules25215092. Molecules. 2020. PMID: 33147885 Free PMC article. Review.
References
-
- Beguin C, Richards MR, Li JG, Wang Y, Xu W, Liu-Chen LY, et al. Synthesis and in vitro evaluation of salvinorin A analogues: effect of configuration at C(2) and substitution at C(18) Bioorg Med Chem Lett. 2006;16:4679–4685. - PubMed
-
- Bolan EA, Kivell B, Jaligam V, Oz M, Jayanthi LD, Han Y, et al. D2 receptors regulate dopamine transporter function via an extracellular signal-regulated kinases 1 and 2-dependent and phosphoinositide 3 kinase-independent mechanism. Mol Pharmacol. 2007;71:1222–1232. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

