iRHOM2-dependent regulation of ADAM17 in cutaneous disease and epidermal barrier function
- PMID: 24643277
- PMCID: PMC4110483
- DOI: 10.1093/hmg/ddu120
iRHOM2-dependent regulation of ADAM17 in cutaneous disease and epidermal barrier function
Abstract
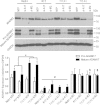
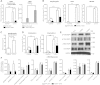
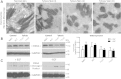
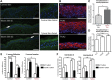
iRHOM2 is a highly conserved, catalytically inactive member of the Rhomboid family, which has recently been shown to regulate the maturation of the multi-substrate ectodomain sheddase enzyme ADAM17 (TACE) in macrophages. Dominant iRHOM2 mutations are the cause of the inherited cutaneous and oesophageal cancer-susceptibility syndrome tylosis with oesophageal cancer (TOC), suggesting a role for this protein in epithelial cells. Here, using tissues derived from TOC patients, we demonstrate that TOC-associated mutations in iRHOM2 cause an increase in the maturation and activity of ADAM17 in epidermal keratinocytes, resulting in significantly upregulated shedding of ADAM17 substrates, including EGF-family growth factors and pro-inflammatory cytokines. This activity is accompanied by increased EGFR activity, increased desmosome processing and the presence of immature epidermal desmosomes, upregulated epidermal transglutaminase activity and heightened resistance to Staphylococcal infection in TOC keratinocytes. Many of these features are consistent with the presence of a constitutive wound-healing-like phenotype in TOC epidermis, which may shed light on a novel pathway in skin repair, regeneration and inflammation.
© The Author 2014. Published by Oxford University Press. All rights reserved. For Permissions, please email: journals.permissions@oup.com.
Figures

References
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

