Alpha/beta interferon receptor signaling amplifies early proinflammatory cytokine production in the lung during respiratory syncytial virus infection
- PMID: 24648449
- PMCID: PMC4093897
- DOI: 10.1128/JVI.00333-14
Alpha/beta interferon receptor signaling amplifies early proinflammatory cytokine production in the lung during respiratory syncytial virus infection
Abstract
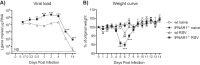
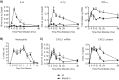
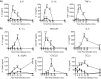
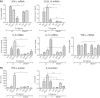
Type I interferons (IFNs) are produced early upon virus infection and signal through the alpha/beta interferon (IFN-α/β) receptor (IFNAR) to induce genes that encode proteins important for limiting viral replication and directing immune responses. To investigate the extent to which type I IFNs play a role in the local regulation of inflammation in the airways, we examined their importance in early lung responses to infection with respiratory syncytial virus (RSV). IFNAR1-deficient (IFNAR1(-/-)) mice displayed increased lung viral load and weight loss during RSV infection. As expected, expression of IFN-inducible genes was markedly reduced in the lungs of IFNAR1(-/-) mice. Surprisingly, we found that the levels of proinflammatory cytokines and chemokines in the lungs of RSV-infected mice were also greatly reduced in the absence of IFNAR signaling. Furthermore, low levels of proinflammatory cytokines were also detected in the lungs of IFNAR1(-/-) mice challenged with noninfectious innate immune stimuli such as selected Toll-like receptor (TLR) agonists. Finally, recombinant IFN-α was sufficient to potentiate the production of inflammatory mediators in the lungs of wild-type mice challenged with innate immune stimuli. Thus, in addition to its well-known role in antiviral resistance, type I IFN receptor signaling acts as a central driver of early proinflammatory responses in the lung. Inhibiting the effects of type I IFNs may therefore be useful in dampening inflammation in lung diseases characterized by enhanced inflammatory cytokine production.
Importance: The initial response to viral infection is characterized by the production of interferons (IFNs). One group of IFNs, the type I IFNs, are produced early upon virus infection and signal through the IFN-α/β receptor (IFNAR) to induce proteins important for limiting viral replication and directing immune responses. Here we examined the importance of type I IFNs in early responses to respiratory syncytial virus (RSV). Our data suggest that type I IFN production and IFNAR receptor signaling not only induce an antiviral state but also serve to amplify proinflammatory responses in the respiratory tract. We also confirm this conclusion in another model of acute inflammation induced by noninfectious stimuli. Our findings are of relevance to human disease, as RSV is a major cause of infant bronchiolitis and polymorphisms in the IFN system are known to impact disease severity.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

