Exogenous albumin inhibits sorafenib-induced cytotoxicity in human cancer cell lines
- PMID: 24649118
- PMCID: PMC3956243
- DOI: 10.3892/mco.2012.3
Exogenous albumin inhibits sorafenib-induced cytotoxicity in human cancer cell lines
Abstract
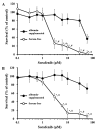
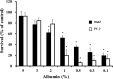
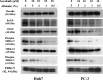
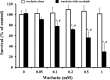
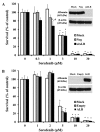
Sorafenib is an orally administered multikinase inhibitor that exhibits anti-angiogenic and anti-tumor activity. Sorafenib is also known to bind to protein (>99.5%), suggesting protein binding may be involved in sorafenib pharmacokinetic variability. Albumin is a major drug-binding protein. In this study, we examined the effect of albumin on sorafenib-induced cytotoxicity using two in vitro culture cell lines, human hepatoma Huh-7 cells and androgen-independent prostate cancer PC-3 cells. The cells were cultured and incubated, and cytotoxicity was assessed. Results were confirmed by western blotting. The presence of exogenous albumin markedly blocked the sorafenib-induced cytotoxicity in the two cell lines. Albumin concentration, the change of pharmacological signal transduction as Raf-B, vascular endothelial growth factor (VEGF), and phosphorylation of MEK1/2 or ERK1/2 were found to be decreased by sorafenib. Co-incubation of warfarin, a representative coumarin anticoagulant and potent binding activity, with albumin enhanced the cytotoxic effects by sorafenib. These mechanisms depend on the high binding proper ties of sorafenib and exogenous albumin. Furthermore, we investigated the effects of endo genous albumin expression on sorafenib-induced cytotoxicity using the siRNA knockdown system or transfected expression vector assay. However, the cytotoxic effects by sorafenib showed little change either with the knockdown or overexpression of albumin. Our results suggest that particular care should be taken with albuminemia or the combined use of drugs with a high affinity for albumin, such as warfarin, and sorafenib in the treatment of cancer patients. Our findings may be useful to the cancer therapeutic strategy by sorafenib.
Keywords: albumin; cytotoxicity; hepatoma; prostate cancer; sorafenib; warfarin.
Figures
Similar articles
-
Sorafenib combined with dasatinib therapy inhibits cell viability, migration, and angiogenesis synergistically in hepatocellular carcinoma.Cancer Chemother Pharmacol. 2021 Jul;88(1):143-153. doi: 10.1007/s00280-021-04272-8. Epub 2021 Apr 16. Cancer Chemother Pharmacol. 2021. PMID: 33860837
-
Sorafenib-induced apoptosis of chronic lymphocytic leukemia cells is associated with downregulation of RAF and myeloid cell leukemia sequence 1 (Mcl-1).Mol Med. 2012 Feb 10;18(1):19-28. doi: 10.2119/molmed.2011.00164. Mol Med. 2012. PMID: 21979753 Free PMC article.
-
Acetylsalicylic Acid Governs the Effect of Sorafenib in RAS-Mutant Cancers.Clin Cancer Res. 2018 Mar 1;24(5):1090-1102. doi: 10.1158/1078-0432.CCR-16-2118. Epub 2017 Dec 1. Clin Cancer Res. 2018. PMID: 29196297 Free PMC article.
-
Sorafenib (BAY 43-9006) in hepatocellular carcinoma patients: from discovery to clinical development.Curr Med Chem. 2012;19(7):938-44. doi: 10.2174/092986712799320736. Curr Med Chem. 2012. PMID: 22214462 Review.
-
Preclinical and clinical development of the oral multikinase inhibitor sorafenib in cancer treatment.Drugs Today (Barc). 2005 Dec;41(12):773-84. doi: 10.1358/dot.2005.41.12.937959. Drugs Today (Barc). 2005. PMID: 16474853 Review.
Cited by
-
Ferroptosis inhibitor alleviates sorafenib-induced cardiotoxicity by attenuating KLF11-mediated FSP1-dependent ferroptosis.Int J Biol Sci. 2024 Apr 22;20(7):2622-2639. doi: 10.7150/ijbs.86479. eCollection 2024. Int J Biol Sci. 2024. PMID: 38725840 Free PMC article.
-
Parenteral Nanoemulsions Loaded with Combined Immuno- and Chemo-Therapy for Melanoma Treatment.Nanomaterials (Basel). 2022 Nov 28;12(23):4233. doi: 10.3390/nano12234233. Nanomaterials (Basel). 2022. PMID: 36500861 Free PMC article.
References
-
- Peters T, editor. All About Albumin: Biochemistry, Genetics, and Medical Applications. Academic Press; San Diego: 1996.
-
- Ballmer PE, McNurlan MA, Milne E, Heys SD, Buchan V, Calder AG, Garlick PJ. Measurement of albumin synthesis in humans: a new approach employing stable isotopes. Am J Physiol. 1990;259:E797–E803. - PubMed
-
- Waldmann TA. Protein-losing enteropathy and kinetic studies of plasma protein metabolism. Semin Nucl Med. 1972;2:251–263. - PubMed
-
- Ballmer PE, Walshe D, McNurlan MA, Watson H, Brunt PW, Garlick PJ. Albumin synthesis rates in cirrhosis: correlation with Child-Turcotte classification. Hepatology. 1993;18:292–297. - PubMed
-
- Pasanisi F, Orban A, Scalfi L, Alfonsi L, Santarpia L, Zurlo E, Celona A, Potenza A, Contaldo F. Predictors of survival in terminal-cancer patients with irreversible bowel obstruction receiving home parenteral nutrition. Nutrition. 2001;17:581–584. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
