Immune privilege of the CNS is not the consequence of limited antigen sampling
- PMID: 24651727
- PMCID: PMC3961746
- DOI: 10.1038/srep04422
Immune privilege of the CNS is not the consequence of limited antigen sampling
Abstract
Central nervous system (CNS) immune privilege is complex, and it is still not understood how CNS antigens are sampled by the peripheral immune system under steady state conditions. To compare antigen sampling from immune-privileged or nonprivileged tissues, we created transgenic mice with oligodendrocyte or gut epithelial cell expression of an EGFP-tagged fusion protein containing ovalbumin (OVA) antigenic peptides and tested peripheral anti-OVA peptide-specific sentinel OT-I and OT-II T cell activation. We report that oligodendrocyte or gut antigens are sampled similarly, as determined by comparable levels of OT-I T cell activation. However, activated T cells do not access the CNS under steady state conditions. These data show that afferent immunity is normally intact as there is no barrier at the antigen sampling level, but that efferent immunity is restricted. To understand how this one-sided surveillance contributes to CNS immune privilege will help us define mechanisms of CNS autoimmune disease initiation.
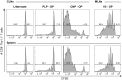
Figures




References
-
- Wekerle H. Breaking ignorance: the case of the brain. Curr. Top. Microbiol. Immunol. 305, 25–50 (2006). - PubMed
-
- Galea I., Bechmann I. & Perry V. H. What is immune privilege (not)? Trends Immunol. 28, 12–18 (2007). - PubMed
-
- Ransohoff R. M. & Engelhardt B. The anatomical and cellular basis of immune surveillance in the central nervous system. Nat. Rev. Immunol. 12, 623–635 (2012). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

