A novel CaM kinase II pathway controls the location of neuropeptide release from Caenorhabditis elegans motor neurons
- PMID: 24653209
- PMCID: PMC3948804
- DOI: 10.1534/genetics.113.158568
A novel CaM kinase II pathway controls the location of neuropeptide release from Caenorhabditis elegans motor neurons
Abstract
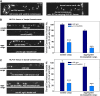
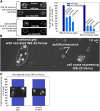
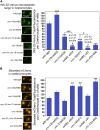
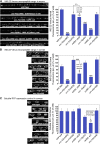
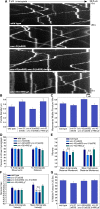
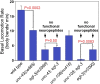
Neurons release neuropeptides via the regulated exocytosis of dense core vesicles (DCVs) to evoke or modulate behaviors. We found that Caenorhabditis elegans motor neurons send most of their DCVs to axons, leaving very few in the cell somas. How neurons maintain this skewed distribution and the extent to which it can be altered to control DCV numbers in axons or to drive release from somas for different behavioral impacts is unknown. Using a forward genetic screen, we identified loss-of-function mutations in UNC-43 (CaM kinase II) that reduce axonal DCV levels by ∼90% and cell soma/dendrite DCV levels by ∼80%, leaving small synaptic vesicles largely unaffected. Blocking regulated secretion in unc-43 mutants restored near wild-type axonal levels of DCVs. Time-lapse video microscopy showed no role for CaM kinase II in the transport of DCVs from cell somas to axons. In vivo secretion assays revealed that much of the missing neuropeptide in unc-43 mutants is secreted via a regulated secretory pathway requiring UNC-31 (CAPS) and UNC-18 (nSec1). DCV cargo levels in unc-43 mutants are similarly low in cell somas and the axon initial segment, indicating that the secretion occurs prior to axonal transport. Genetic pathway analysis suggests that abnormal neuropeptide function contributes to the sluggish basal locomotion rate of unc-43 mutants. These results reveal a novel pathway controlling the location of DCV exocytosis and describe a major new function for CaM kinase II.
Keywords: C. elegans; CaM Kinase II; Dense core vesicle; Neuropeptide release; Regulated secretion; placeholder.
Figures
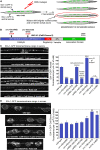
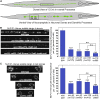
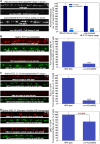
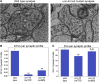

Comment in
-
Dense core vesicle release: controlling the where as well as the when.Genetics. 2014 Mar;196(3):601-4. doi: 10.1534/genetics.113.159905. Genetics. 2014. PMID: 24653208 Free PMC article.
References
-
- Baylis H. A., Furuichi T., Yoshikawa F., Mikoshiba K., Sattelle D. B., 1999. Inositol 1,4,5-trisphosphate receptors are strongly expressed in the nervous system, pharynx, intestine, gonad and excretory cell of Caenorhabditis elegans and are encoded by a single gene (itr-1). J. Mol. Biol. 294: 467–476. - PubMed
-
- Borgonovo B., Ouwendijk J., Solimena M., 2006. Biogenesis of secretory granules. Curr. Opin. Cell Biol. 18: 365–370. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

