To understand muscle you must take it apart
- PMID: 24653704
- PMCID: PMC3949407
- DOI: 10.3389/fphys.2014.00090
To understand muscle you must take it apart
Abstract
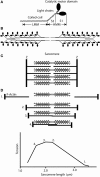
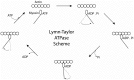
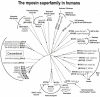
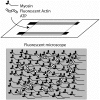
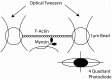
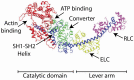
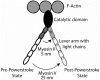
Striated muscle is an elegant system for study at many levels. Much has been learned about the mechanism of contraction from studying the mechanical properties of intact and permeabilized (or skinned) muscle fibers. Structural studies using electron microscopy, X-ray diffraction or spectroscopic probes attached to various contractile proteins were possible because of the highly ordered sarcomeric arrangement of actin and myosin. However, to understand the mechanism of force generation at a molecular level, it is necessary to take the system apart and study the interaction of myosin with actin using in vitro assays. This reductionist approach has lead to many fundamental insights into how myosin powers muscle contraction. In addition, nature has provided scientists with an array of muscles with different mechanical properties and with a superfamily of myosin molecules. Taking advantage of this diversity in myosin structure and function has lead to additional insights into common properties of force generation. This review will highlight the development of the major assays and methods that have allowed this combined reductionist and comparative approach to be so fruitful. This review highlights the history of biochemical and biophysical studies of myosin and demonstrates how a broad comparative approach combined with reductionist studies have led to a detailed understanding of how myosin interacts with actin and uses chemical energy to generate force and movement in muscle contraction and motility in general.
Keywords: ATPase; actomyosin; electron microscopy; in vitro model; muscle; myosin.
Figures

Similar articles
-
Molecular mechanism of actin-myosin motor in muscle.Biochemistry (Mosc). 2011 Dec;76(13):1484-506. doi: 10.1134/S0006297911130086. Biochemistry (Mosc). 2011. PMID: 22339600 Review.
-
Insights into Actin-Myosin Interactions within Muscle from 3D Electron Microscopy.Int J Mol Sci. 2019 Apr 5;20(7):1703. doi: 10.3390/ijms20071703. Int J Mol Sci. 2019. PMID: 30959804 Free PMC article. Review.
-
Regulation of contraction in striated muscle.Physiol Rev. 2000 Apr;80(2):853-924. doi: 10.1152/physrev.2000.80.2.853. Physiol Rev. 2000. PMID: 10747208 Review.
-
Molecular mechanism of ATP-dependent actin-myosin interaction in muscle contraction.Jpn J Physiol. 1993;43(4):435-54. doi: 10.2170/jjphysiol.43.435. Jpn J Physiol. 1993. PMID: 8114356 Review.
-
Special Issue: The Actin-Myosin Interaction in Muscle: Background and Overview.Int J Mol Sci. 2019 Nov 14;20(22):5715. doi: 10.3390/ijms20225715. Int J Mol Sci. 2019. PMID: 31739584 Free PMC article. Review.
Cited by
-
Alterations of the skeletal muscle contractile apparatus in necrosis induced by myotoxic snake venom phospholipases A2: a mini-review.J Muscle Res Cell Motil. 2024 Jun;45(2):69-77. doi: 10.1007/s10974-023-09662-4. Epub 2023 Dec 8. J Muscle Res Cell Motil. 2024. PMID: 38063951 Free PMC article. Review.
-
Force Measurements From Myofibril to Filament.Front Physiol. 2022 Jan 27;12:817036. doi: 10.3389/fphys.2021.817036. eCollection 2021. Front Physiol. 2022. PMID: 35153821 Free PMC article. Review.
-
Omics Strategies in Current Advancements of Infectious Fish Disease Management.Biology (Basel). 2021 Oct 22;10(11):1086. doi: 10.3390/biology10111086. Biology (Basel). 2021. PMID: 34827079 Free PMC article. Review.
-
Myocardial Contractility: Historical and Contemporary Considerations.Front Physiol. 2020 Mar 31;11:222. doi: 10.3389/fphys.2020.00222. eCollection 2020. Front Physiol. 2020. PMID: 32296340 Free PMC article.
-
The effects of inorganic phosphate on muscle force development and energetics: challenges in modelling related to experimental uncertainties.J Muscle Res Cell Motil. 2021 Mar;42(1):33-46. doi: 10.1007/s10974-019-09558-2. Epub 2019 Oct 16. J Muscle Res Cell Motil. 2021. PMID: 31620962 Free PMC article.
References
-
- Banga I. (1942). The phosphatase activity of myosin. Stud. Inst. Med. Chem. Univ. Szeged. 1, 27–36
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

