Programming nanopore ion flow for encoded multiplex microRNA detection
- PMID: 24654890
- PMCID: PMC4004327
- DOI: 10.1021/nn406339n
Programming nanopore ion flow for encoded multiplex microRNA detection
Abstract
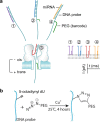
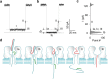
Many efforts are being made in translating the nanopore into an ultrasensitive single-molecule platform for various genetic and epigenetic detections. However, compared with current approaches including PCR, the low throughput limits the nanopore applications in biological research and clinical settings, which usually requires simultaneous detection of multiple biomarkers for accurate disease diagnostics. Herein we report a barcode probe approach for multiple nucleic acid detection in one nanopore. Instead of directly identifying different targets in a nanopore, we designed a series of barcode probes to encode different targets. When the probe is bound with the target, the barcode group polyethylene glycol attached on the probe through click chemistry can specifically modulate nanopore ion flow. The resulting signature serves as a marker for the encoded target. Therefore counting different signatures in a current recording allows simultaneous analysis of multiple targets in one nanopore. The principle of this approach was verified by using a panel of cancer-derived microRNAs as the target, a type of biomarker for cancer detection.
Figures


Similar articles
-
Nanopore single-molecule dielectrophoretic detection of cancer-derived microRNA biomarkers.Annu Int Conf IEEE Eng Med Biol Soc. 2013;2013:6821-4. doi: 10.1109/EMBC.2013.6611124. Annu Int Conf IEEE Eng Med Biol Soc. 2013. PMID: 24111311 Free PMC article.
-
Digital counting of nucleic acid targets using solid-state nanopores.Nanoscale. 2020 Sep 14;12(34):17833-17840. doi: 10.1039/d0nr03878d. Epub 2020 Aug 24. Nanoscale. 2020. PMID: 32832949
-
Rapid electronic detection of probe-specific microRNAs using thin nanopore sensors.Nat Nanotechnol. 2010 Nov;5(11):807-14. doi: 10.1038/nnano.2010.202. Epub 2010 Oct 24. Nat Nanotechnol. 2010. PMID: 20972437
-
Detection of miRNAs with a nanopore single-molecule counter.Expert Rev Mol Diagn. 2012 Jul;12(6):573-84. doi: 10.1586/erm.12.58. Expert Rev Mol Diagn. 2012. PMID: 22845478 Free PMC article. Review.
-
Chemistry solutions to facilitate nanopore detection and analysis.Biosens Bioelectron. 2022 Oct 1;213:114448. doi: 10.1016/j.bios.2022.114448. Epub 2022 Jun 1. Biosens Bioelectron. 2022. PMID: 35716643 Review.
Cited by
-
Characterization of Interstrand DNA-DNA Cross-Links Using the α-Hemolysin Protein Nanopore.ACS Nano. 2015 Dec 22;9(12):11812-9. doi: 10.1021/acsnano.5b03923. Epub 2015 Nov 18. ACS Nano. 2015. PMID: 26563913 Free PMC article.
-
Polycationic Probe-Guided Nanopore Single-Molecule Counter for Selective miRNA Detection.Methods Mol Biol. 2017;1632:255-268. doi: 10.1007/978-1-4939-7138-1_17. Methods Mol Biol. 2017. PMID: 28730445 Free PMC article.
-
Probing molecular pathways for DNA orientational trapping, unzipping and translocation in nanopores by using a tunable overhang sensor.Nanoscale. 2014 Oct 7;6(19):11372-9. doi: 10.1039/c4nr03195d. Nanoscale. 2014. PMID: 25144935 Free PMC article.
-
Mimicking Ribosomal Unfolding of RNA Pseudoknot in a Protein Channel.J Am Chem Soc. 2015 Dec 23;137(50):15742-52. doi: 10.1021/jacs.5b07910. Epub 2015 Dec 10. J Am Chem Soc. 2015. PMID: 26595106 Free PMC article.
-
Nanopore electric snapshots of an RNA tertiary folding pathway.Nat Commun. 2017 Nov 13;8(1):1458. doi: 10.1038/s41467-017-01588-z. Nat Commun. 2017. PMID: 29133841 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

