A mild, ferrocene-catalyzed C-H imidation of (hetero)arenes
- PMID: 24654983
- PMCID: PMC4210156
- DOI: 10.1021/ja501879c
A mild, ferrocene-catalyzed C-H imidation of (hetero)arenes
Abstract
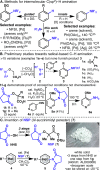
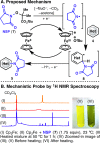
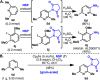
A simple method for direct C-H imidation is reported using a new perester-based self-immolating reagent and a base-metal catalyst. The succinimide products obtained can be easily deprotected in situ (if desired) to reveal the corresponding anilines directly. The scope of the reaction is broad, the conditions are extremely mild, and the reaction is tolerant of oxidizable and acid-labile functionality, multiple heteroatoms, and aryl iodides. Mechanistic studies indicate that ferrocene (Cp2Fe) plays the role of an electron shuttle in the decomposition of the perester reagent, delivering a succinimidyl radical ready to add to an aromatic system.
Figures
References
-
-
For intramolecular (hetero)arene C–H amination, see:
- Tsang W. C. P.; Zheng N.; Buchwald S. L. J. Am. Chem. Soc. 2005, 127, 14560. - PubMed
- Tan Y.; Hartwig J. F. J. Am. Chem. Soc. 2010, 132, 3676. - PubMed
- Engle K. M.; Mei T.-S.; Wang X.; Yu J.-Q. Angew. Chem., Int. Ed. 2011, 50, 1478. - PMC - PubMed
- Jordan-Hore J. A.; Johansson C. C. C.; Gulia M.; Beck E. M.; Gaunt M. J. J. Am. Chem. Soc. 2008, 130, 16184. - PubMed
- Wang H.; Wang Y.; Peng C.; Zhang J.; Zhu Q. J. Am. Chem. Soc. 2010, 132, 13217. - PubMed
- Antonchick A. P.; Samanta R.; Kulikov K.; Lategahn J. Angew. Chem., Int. Ed. 2011, 50, 8605. - PubMed
- King A. E.; Huffman L. M.; Casitas A.; Costas M.; Ribas X.; Stahl S. S. J. Am. Chem. Soc. 2010, 132, 12068. - PubMed
- Brasche G.; Buchwald S. L. Angew. Chem., Int. Ed. 2008, 47, 1932. - PubMed
-
For reviews on (hetero)arene C–H aminations, see:
- Hartwig J. F. Acc. Chem. Res. 2012, 45, 864. - PubMed
- Mueller P.; Fruit C. Chem. Rev. 2003, 103, 2905. - PubMed
- Dick A. R.; Sanford M. S. Tetrahedron 2006, 62, 2439.
- Davies H. M. L.; Long M. S. Angew. Chem., Int. Ed. 2005, 44, 3518. - PubMed
- Davies H. M. L.; Manning J. R. Nature 2008, 451, 417. - PMC - PubMed
- Cho S. H.; Kim J. Y.; Kwak J. Y.; Kwak J.; Chang S. Chem. Soc. Rev. 2011, 40, 5068. - PubMed
-
-
- Brückl T.; Baxter R. D.; Ishihara Y.; Baran P. S. Acc. Chem. Res. 2012, 45, 826. - PMC - PubMed
-
For guided (hetero)arene C–H amination, see:
- Shang M.; Zeng S.-H.; Sun S.-Z.; Dai H.-X.; Yu J.-Q. Org. Lett. 2013, 15, 5286. - PubMed
- Yoo E. J.; Ma S.; Mei T.-S.; Chan K. S. L.; Yu J.-Q. J. Am. Chem. Soc. 2011, 133, 7652. - PubMed
- Shang M.; Sun S.-Z.; Dai H.-X.; Yu J.-Q. J. Am. Chem. Soc. 2014, 136, 3354. - PubMed
- Sun K.; Li Y.; Xiong T.; Zhang J.; Zhang Q. J. Am. Chem. Soc. 2011, 133, 1694. - PubMed
- Thu H.-Y.; Yu W.-Y.; Che C.-M. J. Am. Chem. Soc. 2006, 128, 9048. - PubMed
- Ng K.-H.; Chan A. S. C.; Yu W.-Y. J. Am. Chem. Soc. 2010, 132, 12862. - PubMed
- Dick A. R.; Remy M. S.; Kampf J. W.; Sanford M. S. Organometallics 2007, 26, 1365.
- Matsubara T.; Asako S.; Ilies L.; Nakamura E. J. Am. Chem. Soc. 2014, 136, 646. - PubMed
- Tran L. D.; Roane J.; Daugulis O. Angew. Chem., Int. Ed. 2013, 52, 6043. - PMC - PubMed
-
For innate (hetero)arene intermolecular C–H amination, see:
- Kim H. J.; Kim J.; Cho S. H.; Chang S. J. Am. Chem. Soc. 2011, 133, 16382. - PubMed
- Kantak A. A.; Potavathri S.; Barham R. A.; Romano K. M.; DeBoef B. J. Am. Chem. Soc. 2011, 133, 19960. - PMC - PubMed
- Shrestha R.; Mukherjee P.; Tan Y.; Litman Z. C.; Hartwig J. F. J. Am. Chem. Soc. 2013, 135, 8480. - PubMed
- Boursalian G. B.; Ngai M.-Y.; Hojczyk K. N.; Ritter T. J. Am. Chem. Soc. 2013, 135, 13278. - PMC - PubMed
- Kawano T.; Hirano K.; Satoh T.; Miura M. J. Am. Chem. Soc. 2010, 132, 6900. - PubMed
- Wang Q.; Schreiber S. L. Org. Lett. 2009, 11, 5178. - PMC - PubMed
- Monguchi D.; Fujiwara T.; Furukawa H.; Mori A. Org. Lett. 2009, 11, 1607. - PubMed
- Citterio A.; Gentile A.; Minisci F.; Navarrini V.; Serravalle M.; Ventura S. J. Org. Chem. 1984, 49, 4479.
- Lidgett R. A.; Lynch E. R.; McCall E. B. J. Chem. Soc. 1965, 3754.
- Cadogan J. I. G.; Rowley A. G. J. Chem. Soc., Perkin Trans. 1 1975, 1069.
- Day J. C.; Katsaros M. G.; Kocher W. D.; Scott A. E.; Skell P. S. J. Am. Chem. Soc. 1978, 100, 1950.
- Lu F.-L.; Naguib Y. M. A.; Kitadani M.; Chow Y. L. Can. J. Chem. 1979, 57, 1967.
- Togo H.; Hoshina Y.; Muraki T.; Nakayama H.; Yokoyama M. J. Org. Chem. 1998, 63, 5193.
-
- Zard S. Z. Chem. Soc. Rev. 2008, 37, 1603. - PubMed
- Danen W. C.; Neugebauer F. A. Angew. Chem., Int. Ed. 1975, 14, 783.
- Stella L. Angew. Chem., Int. Ed. 1983, 22, 337.
-
- Forrester A. R.; Gill M.; Meyer C. J.; Sadd J. S.; Thomson R. H. J. Chem. Soc., Chem. Commun. 1975, 291.
- Boivin J.; Fouquet E.; Zard S. Z. Tetrahedron 1994, 50, 1769.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

