Tumor necrosis factor-α impairs oligodendroglial differentiation through a mitochondria-dependent process
- PMID: 24658399
- PMCID: PMC4085526
- DOI: 10.1038/cdd.2014.35
Tumor necrosis factor-α impairs oligodendroglial differentiation through a mitochondria-dependent process
Abstract
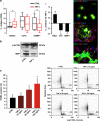
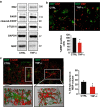
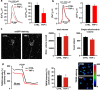
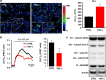
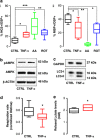
Mitochondrial defects, affecting parameters such as mitochondrial number and shape, levels of respiratory chain complex components and markers of oxidative stress, have been associated with the appearance and progression of multiple sclerosis. Nevertheless, mitochondrial physiology has never been monitored during oligodendrocyte progenitor cell (OPC) differentiation, especially in OPCs challenged with proinflammatory cytokines. Here, we show that tumor necrosis factor alpha (TNF-α) inhibits OPC differentiation, accompanied by altered mitochondrial calcium uptake, mitochondrial membrane potential, and respiratory complex I activity as well as increased reactive oxygen species production. Treatment with a mitochondrial uncoupler (FCCP) to mimic mitochondrial impairment also causes cells to accumulate at the progenitor stage. Interestingly, AMP-activated protein kinase (AMPK) levels increase during TNF-α exposure and inhibit OPC differentiation. Overall, our data indicate that TNF-α induces metabolic changes, driven by mitochondrial impairment and AMPK activation, leading to the inhibition of OPC differentiation.
Figures

References
-
- Ghafourifar P, Mousavizadeh K, Parihar MS, Nazarewicz RR, Parihar A, Zenebe WJ. Mitochondria in multiple sclerosis. Front Biosci. 2008;13:3116–3126. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

