TDP-43 is a key player in the clinical features associated with Alzheimer's disease
- PMID: 24659241
- PMCID: PMC4172544
- DOI: 10.1007/s00401-014-1269-z
TDP-43 is a key player in the clinical features associated with Alzheimer's disease
Abstract
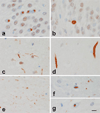
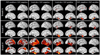
The aim of this study was to determine whether the TAR DNA-binding protein of 43 kDa (TDP-43) has any independent effect on the clinical and neuroimaging features typically ascribed to Alzheimer's disease (AD) pathology, and whether TDP-43 pathology could help shed light on the phenomenon of resilient cognition in AD. Three-hundred and forty-two subjects pathologically diagnosed with AD were screened for the presence, burden and distribution of TDP-43. All had been classified as cognitively impaired or normal, prior to death. Atlas-based parcellation and voxel-based morphometry were used to assess regional atrophy on MRI. Regression models controlling for age at death, apolipoprotein ε4 and other AD-related pathologies were utilized to explore associations between TDP-43 and cognition or brain atrophy, stratified by Braak stage. In addition, we determined whether the effects of TDP-43 were mediated by hippocampal sclerosis. One-hundred and ninety-five (57%) cases were TDP-positive. After accounting for age, apolipoprotein ε4 and other pathologies, TDP-43 had a strong effect on cognition, memory loss and medial temporal atrophy in AD. These effects were not mediated by hippocampal sclerosis. TDP-positive subjects were 10× more likely to be cognitively impaired at death compared to TDP-negative subjects. Greater cognitive impairment and medial temporal atrophy were associated with greater TDP-43 burden and more extensive TDP-43 distribution. TDP-43 is an important factor in the manifestation of the clinico-imaging features of AD. TDP-43 also appears to be able to overpower what has been termed resilient brain aging. TDP-43 therefore should be considered a potential therapeutic target for the treatment of AD.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures





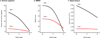
References
-
- Agresti A. Categorical Data Analysis. City: Wiley-Interscience; 2002.
-
- Alzheimer A. Über eine eigenartige Erkrankung der Hirnrinde. Allgemeine Zeitschrift für Psychiatrie und Psychisch-Gerichtliche Medizin. 1907;64:146–148.
-
- Arai T, Mackenzie IR, Hasegawa M, et al. Phosphorylated TDP-43 in Alzheimer's disease and dementia with Lewy bodies. Acta neuropathologica. 2009;117:125–136. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

