ADAM10 is the major sheddase responsible for the release of membrane-associated meprin A
- PMID: 24662289
- PMCID: PMC4036340
- DOI: 10.1074/jbc.M114.559088
ADAM10 is the major sheddase responsible for the release of membrane-associated meprin A
Abstract
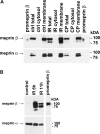
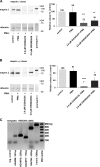
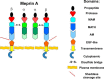
Meprin A, composed of α and β subunits, is a membrane-bound metalloproteinase in renal proximal tubules. Meprin A plays an important role in tubular epithelial cell injury during acute kidney injury (AKI). The present study demonstrated that during ischemia-reperfusion-induced AKI, meprin A was shed from proximal tubule membranes, as evident from its redistribution toward the basolateral side, proteolytic processing in the membranes, and excretion in the urine. To identify the proteolytic enzyme responsible for shedding of meprin A, we generated stable HEK cell lines expressing meprin β alone and both meprin α and meprin β for the expression of meprin A. Phorbol 12-myristate 13-acetate and ionomycin stimulated ectodomain shedding of meprin β and meprin A. Among the inhibitors of various proteases, the broad spectrum inhibitor of the ADAM family of proteases, tumor necrosis factor-α protease inhibitor (TAPI-1), was most effective in preventing constitutive, phorbol 12-myristate 13-acetate-, and ionomycin-stimulated shedding of meprin β and meprin A in the medium of both transfectants. The use of differential inhibitors for ADAM10 and ADAM17 indicated that ADAM10 inhibition is sufficient to block shedding. In agreement with these results, small interfering RNA to ADAM10 but not to ADAM9 or ADAM17 inhibited meprin β and meprin A shedding. Furthermore, overexpression of ADAM10 resulted in enhanced shedding of meprin β from both transfectants. Our studies demonstrate that ADAM10 is the major ADAM metalloproteinase responsible for the constitutive and stimulated shedding of meprin β and meprin A. These studies further suggest that inhibiting ADAM 10 activity could be of therapeutic benefit in AKI.
Keywords: ADAM ADAMTS; ADAM10; ADAM17; Kidney; Meprin; Metalloprotease; Mouse; Shedding; siRNA.
Figures






Similar articles
-
Role of ADAM10 and ADAM17 in CD16b shedding mediated by different stimulators.Chin Med Sci J. 2012 Jun;27(2):73-9. Chin Med Sci J. 2012. PMID: 22770404
-
Meprin β induces activities of A disintegrin and metalloproteinases 9, 10, and 17 by specific prodomain cleavage.FASEB J. 2019 Nov;33(11):11925-11940. doi: 10.1096/fj.201801371R. Epub 2019 Aug 9. FASEB J. 2019. PMID: 31381863 Free PMC article.
-
Identification of ADAM10 as a major TNF sheddase in ADAM17-deficient fibroblasts.Cytokine. 2009 Jun;46(3):309-15. doi: 10.1016/j.cyto.2009.03.002. Epub 2009 Apr 5. Cytokine. 2009. PMID: 19346138
-
Metalloproteinase inhibitors for the disintegrin-like metalloproteinases ADAM10 and ADAM17 that differentially block constitutive and phorbol ester-inducible shedding of cell surface molecules.Comb Chem High Throughput Screen. 2005 Mar;8(2):161-71. doi: 10.2174/1386207053258488. Comb Chem High Throughput Screen. 2005. PMID: 15777180 Review.
-
The "A Disintegrin And Metalloproteases" ADAM10 and ADAM17: novel drug targets with therapeutic potential?Eur J Cell Biol. 2011 Jun-Jul;90(6-7):527-35. doi: 10.1016/j.ejcb.2010.11.005. Epub 2010 Dec 30. Eur J Cell Biol. 2011. PMID: 21194787 Review.
Cited by
-
PLD1 promotes dendritic spine development by inhibiting ADAM10-mediated N-cadherin cleavage.Sci Rep. 2017 Jul 20;7(1):6035. doi: 10.1038/s41598-017-06121-2. Sci Rep. 2017. PMID: 28729535 Free PMC article.
-
The MUC2 Gene Product: Polymerisation and Post-Secretory Organisation-Current Models.Polymers (Basel). 2024 Jun 12;16(12):1663. doi: 10.3390/polym16121663. Polymers (Basel). 2024. PMID: 38932019 Free PMC article. Review.
-
Differences in Shedding of the Interleukin-11 Receptor by the Proteases ADAM9, ADAM10, ADAM17, Meprin α, Meprin β and MT1-MMP.Int J Mol Sci. 2019 Jul 26;20(15):3677. doi: 10.3390/ijms20153677. Int J Mol Sci. 2019. PMID: 31357561 Free PMC article.
-
Cell adhesion molecule-1 shedding induces apoptosis of renal epithelial cells and exacerbates human nephropathies.Am J Physiol Renal Physiol. 2018 Mar 1;314(3):F388-F398. doi: 10.1152/ajprenal.00385.2017. Epub 2017 Oct 25. Am J Physiol Renal Physiol. 2018. PMID: 29070574 Free PMC article.
-
Contribution of ADAM17 and related ADAMs in cardiovascular diseases.Cell Mol Life Sci. 2021 May;78(9):4161-4187. doi: 10.1007/s00018-021-03779-w. Epub 2021 Feb 11. Cell Mol Life Sci. 2021. PMID: 33575814 Free PMC article. Review.
References
-
- Becker-Pauly C., Höwel M., Walker T., Vlad A., Aufenvenne K., Oji V., Lottaz D., Sterchi E. E., Debela M., Magdolen V., Traupe H., Stöcker W. (2007) The α and β subunits of the metalloprotease meprin are expressed in separate layers of human epidermis, revealing different functions in keratinocyte proliferation and differentiation. J. Invest. Dermatol. 127, 1115–1125 - PubMed
-
- Lottaz D., Hahn D., Müller S., Müller C., Sterchi E. E. (1999) Secretion of human meprin from intestinal epithelial cells depends on differential expression of the α and β subunits. Eur. J. Biochem. 259, 496–504 - PubMed
-
- Lottaz D., Maurer C. A., Hahn D., Büchler M. W., Sterchi E. E. (1999) Nonpolarized secretion of human meprin α in colorectal cancer generates an increased proteolytic potential in the stroma. Cancer Res. 59, 1127–1133 - PubMed
-
- Crisman J. M., Zhang B., Norman L. P., Bond J. S. (2004) Deletion of the mouse meprin β metalloprotease gene diminishes the ability of leukocytes to disseminate through extracellular matrix. J. Immunol. 172, 4510–4519 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

