Discharge profiles across the sleep-waking cycle of identified cholinergic, GABAergic, and glutamatergic neurons in the pontomesencephalic tegmentum of the rat
- PMID: 24672016
- PMCID: PMC3965793
- DOI: 10.1523/JNEUROSCI.2617-13.2014
Discharge profiles across the sleep-waking cycle of identified cholinergic, GABAergic, and glutamatergic neurons in the pontomesencephalic tegmentum of the rat
Abstract
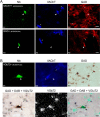
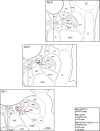
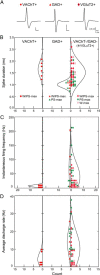
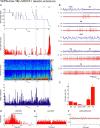
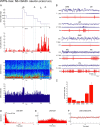
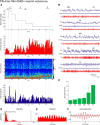
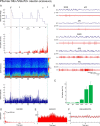
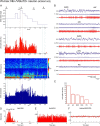
Distributed within the laterodorsal tegmental and pedunculopontine tegmental nuclei (LDT and PPT), cholinergic neurons in the pontomesencephalic tegmentum have long been thought to play a critical role in stimulating cortical activation during waking (W) and paradoxical sleep (PS, also called REM sleep), yet also in promoting PS with muscle atonia. However, the discharge profile and thus precise roles of the cholinergic neurons have remained uncertain because they lie intermingled with GABAergic and glutamatergic neurons, which might also assume these roles. By applying juxtacellular recording and labeling in naturally sleeping-waking, head-fixed rats, we investigated the discharge profiles of histochemically identified cholinergic, GABAergic, and glutamatergic neurons in the LDT, SubLDT, and adjoining medial part of the PPT (MPPT) in relation to sleep-wake states, cortical activity, and muscle tone. We found that all cholinergic neurons were maximally active during W and PS in positive correlation with fast (γ) cortical activity, as "W/PS-max active neurons." Like cholinergic neurons, many GABAergic and glutamatergic neurons were also "W/PS-max active." Other GABAergic and glutamatergic neurons were "PS-max active," being minimally active during W and maximally active during PS in negative correlation with muscle tone. Conversely, some glutamatergic neurons were "W-max active," being maximally active during W and minimally active during PS in positive correlation with muscle tone. Through different discharge profiles, the cholinergic, GABAergic, and glutamatergic neurons of the LDT, SubLDT, and MPPT thus appear to play distinct roles in promoting W and PS with cortical activation, PS with muscle atonia, or W with muscle tone.
Keywords: EEG; EMG; REM sleep; paradoxical sleep; slow-wave sleep.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
