Diabetic retinopathy: loss of neuroretinal adaptation to the diabetic metabolic environment
- PMID: 24673341
- PMCID: PMC4154702
- DOI: 10.1111/nyas.12412
Diabetic retinopathy: loss of neuroretinal adaptation to the diabetic metabolic environment
Abstract
Diabetic retinopathy (DR) impairs vision of patients with type 1 and type 2 diabetes, associated with vascular dysfunction and occlusion, retinal edema, hemorrhage, and inappropriate growth of new blood vessels. The recent success of biologic treatments targeting vascular endothelial growth factor (VEGF) demonstrates that treating the vascular aspects in the later stages of the disease can preserve vision in many patients. It would also be highly desirable to prevent the onset of the disease or arrest its progression at a stage preceding the appearance of overt microvascular pathologies. The progression of DR is not necessarily linear but may follow a series of steps that evolve over the course of multiple years. Abundant data suggest that diabetes affects the entire neurovascular unit of the retina, with an early loss of neurovascular coupling, gradual neurodegeneration, gliosis, and neuroinflammation occurring before observable vascular pathologies. In this article, we consider the pathology of DR from the point of view that diabetes causes measurable dysfunctions in the complex integral network of cell types that produce and maintain human vision.
Keywords: diabetic retinopathy; maladaption; metabolism; neurodegeneration; neurovascular unit.
© 2014 New York Academy of Sciences.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

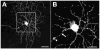
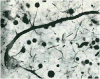



References
-
- Pascolini D, Mariotti SP. Global estimates of visual impairment: 2010. Br J Ophthalmol. 2012;96:614–618. doi:610.1136/bjophthalmol-2011-300539 Epub 302011 Dec 300531. - PubMed
-
- Sivaprasad S, et al. Prevalence of diabetic retinopathy in various ethnic groups: a worldwide perspective. Surv Ophthalmol. 2012;57:347–370. Epub 2012 Apr 2028. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

