Effect of silica particle size on macrophage inflammatory responses
- PMID: 24681489
- PMCID: PMC3969333
- DOI: 10.1371/journal.pone.0092634
Effect of silica particle size on macrophage inflammatory responses
Abstract
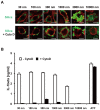
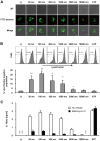
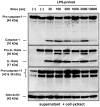
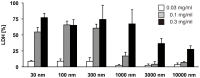
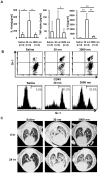
Amorphous silica particles, such as nanoparticles (<100 nm diameter particles), are used in a wide variety of products, including pharmaceuticals, paints, cosmetics, and food. Nevertheless, the immunotoxicity of these particles and the relationship between silica particle size and pro-inflammatory activity are not fully understood. In this study, we addressed the relationship between the size of amorphous silica (particle dose, diameter, number, and surface area) and the inflammatory activity (macrophage phagocytosis, inflammasome activation, IL-1β secretion, cell death and lung inflammation). Irrespective of diameter size, silica particles were efficiently internalized by mouse bone marrow-derived macrophages via an actin cytoskeleton-dependent pathway, and induced caspase-1, but not caspase-11, activation. Of note, 30 nm-1000 nm diameter silica particles induced lysosomal destabilization, cell death, and IL-1β secretion at markedly higher levels than did 3000 nm-10000 nm silica particles. Consistent with in vitro results, intra-tracheal administration of 30 nm silica particles into mice caused more severe lung inflammation than that of 3000 nm silica particles, as assessed by measurement of pro-inflammatory cytokines and neutrophil infiltration in bronchoalveolar lavage fluid of mice, and by the micro-computed tomography analysis. Taken together, these results suggest that silica particle size impacts immune responses, with submicron amorphous silica particles inducing higher inflammatory responses than silica particles over 1000 nm in size, which is ascribed not only to their ability to induce caspase-1 activation but also to their cytotoxicity.
Conflict of interest statement
Figures
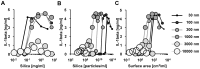
Similar articles
-
Comparison of non-crystalline silica nanoparticles in IL-1β release from macrophages.Part Fibre Toxicol. 2012 Aug 10;9:32. doi: 10.1186/1743-8977-9-32. Part Fibre Toxicol. 2012. PMID: 22882971 Free PMC article.
-
The effect of surface modification of amorphous silica particles on NLRP3 inflammasome mediated IL-1beta production, ROS production and endosomal rupture.Biomaterials. 2010 Sep;31(26):6833-42. doi: 10.1016/j.biomaterials.2010.05.036. Epub 2010 Jun 18. Biomaterials. 2010. PMID: 20561679
-
The alarmin IL-1α is a master cytokine in acute lung inflammation induced by silica micro- and nanoparticles.Part Fibre Toxicol. 2014 Dec 13;11:69. doi: 10.1186/s12989-014-0069-x. Part Fibre Toxicol. 2014. PMID: 25497724 Free PMC article.
-
The complex cascade of cellular events governing inflammasome activation and IL-1β processing in response to inhaled particles.Part Fibre Toxicol. 2016 Aug 12;13(1):40. doi: 10.1186/s12989-016-0150-8. Part Fibre Toxicol. 2016. PMID: 27519871 Free PMC article. Review.
-
Centrality of Myeloid-Lineage Phagocytes in Particle-Triggered Inflammation and Autoimmunity.Front Toxicol. 2021 Nov 4;3:777768. doi: 10.3389/ftox.2021.777768. eCollection 2021. Front Toxicol. 2021. PMID: 35295146 Free PMC article. Review.
Cited by
-
In Vitro Determination of the Immunogenic Impact of Nanomaterials on Primary Peripheral Blood Mononuclear Cells.Int J Mol Sci. 2020 Aug 5;21(16):5610. doi: 10.3390/ijms21165610. Int J Mol Sci. 2020. PMID: 32764386 Free PMC article.
-
Microfluidic-assisted silk nanoparticle tuning.Nanoscale Adv. 2018 Nov 30;1(2):873-883. doi: 10.1039/c8na00208h. eCollection 2019 Feb 12. Nanoscale Adv. 2018. PMID: 36132231 Free PMC article.
-
Biological Effects of Nanoparticles on Macrophage Polarization in the Tumor Microenvironment.Nanotheranostics. 2019 Jan 1;3(1):66-88. doi: 10.7150/ntno.30052. eCollection 2019. Nanotheranostics. 2019. PMID: 30662824 Free PMC article. Review.
-
A pathologic two-way street: how innate immunity impacts lung fibrosis and fibrosis impacts lung immunity.Clin Transl Immunology. 2019 Jun 26;8(6):e1065. doi: 10.1002/cti2.1065. eCollection 2019. Clin Transl Immunology. 2019. PMID: 31293783 Free PMC article. Review.
-
A novel Approach for Non-Invasive Lung Imaging and Targeting Lung Immune Cells.Int J Mol Sci. 2020 Feb 27;21(5):1613. doi: 10.3390/ijms21051613. Int J Mol Sci. 2020. PMID: 32120819 Free PMC article.
References
-
- Leung CC, Yu ITS, Chen WH (2012) Silicosis. Lancet 379: 2008–2018. - PubMed
-
- WHO (2007) The Global Occupational Health Network newsletter: elimination of silicosis. http://www.who.int/occupational_health/publications/newsletter/gohnet12e.... Issue No. 12 – 2007
-
- Bowman DM, van Calster G, Friedrichs S (2010) Nanomaterials and regulation of cosmetics. Nat Nanotechnol 5: 92–92. - PubMed
-
- Kaewamatawong T, Kawamura N, Okajima M, Sawada M, Morita T, et al. (2005) Acute pulmonary toxicity caused by exposure to colloidal silica: Particle size dependent pathological changes in mice. Toxicol Pathol 33: 743–749. - PubMed
-
- Cho W-S, Choi M, Han BS, Cho M, Oh J, et al. (2007) Inflammatory mediators induced by intratracheal instillation of ultrafine amorphous silica particles. Toxicol Lett 175: 24–33. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

