Mechanistic target of rapamycin complex 1 expands Th17 and IL-4+ CD4-CD8- double-negative T cells and contracts regulatory T cells in systemic lupus erythematosus
- PMID: 24683191
- PMCID: PMC3995867
- DOI: 10.4049/jimmunol.1301859
Mechanistic target of rapamycin complex 1 expands Th17 and IL-4+ CD4-CD8- double-negative T cells and contracts regulatory T cells in systemic lupus erythematosus
Abstract
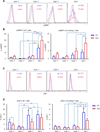
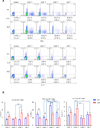
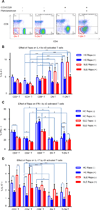
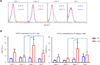
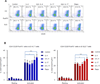
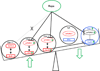
The mechanistic target of rapamycin (mTOR) is activated in CD4(-)CD8(-) double-negative (DN) T cells and its blockade is therapeutic in systemic lupus erythematosus (SLE) patients. Murine studies showed the involvement of mTOR complex 1 (mTORC1) and 2 (mTORC2) in the differentiation of Th1/Th17 cells and Th2 cells, respectively. In this study, we investigated the roles of mTORC1 and mTORC2 in T cell lineage development in SLE and matched healthy control (HC) subjects. mTORC1 activity was increased, whereas mTORC2 was reduced, as assessed by phosphorylation of their substrates phosphorylated S6 kinase 1 or phosphorylated S6 ribosomal protein and phosphorylated Akt, respectively. Rapamycin inhibited mTORC1 and enhanced mTORC2. IL-4 expression was increased in freshly isolated CD8(+) lupus T cells (SLE: 8.09 ± 1.93%, HC: 3.61 ± 0.49%; p = 0.01). DN T cells had greater IL-4 expression than CD4(+) or CD8(+) T cells of SLE patients after 3-d in vitro stimulation, which was suppressed by rapamycin (control: 9.26 ± 1.48%, rapamycin: 5.03 ± 0.66%; p < 0.001). GATA-3 expression was increased in CD8(+) lupus T cells (p < 0.01) and was insensitive to rapamycin treatment. IFN-γ expression was reduced in all lupus T cell subsets (p = 1.0 × 10(-5)) and also resisted rapamycin. IL-17 expression was increased in CD4(+) lupus T cells (SLE: 3.62 ± 0.66%, HC: 2.29 ± 0.27%; p = 0.019), which was suppressed by rapamycin (control: 3.91 ± 0.79%, rapamycin: 2.22 ± 0.60%; p < 0.001). Frequency of regulatory T cells (Tregs) was reduced in SLE (SLE: 1.83 ± 0.25%, HC: 2.97 ± 0.27%; p = 0.0012). Rapamycin inhibited mTORC1 in Tregs and promoted their expansion. Neutralization of IL-17, but not IL-4, also expanded Tregs in SLE and HC subjects. These results indicate that mTORC1 expands IL-4(+) DN T and Th17 cells, and contracts Tregs in SLE.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures


References
-
- Shivakumar S, Tsokos GC, Datta SK. T cell receptor alpha/beta expressing double-negative (CD4−/CD8−) and CD4+ T helper cells in humans augment the production of pathogenic anti-DNA autoantibodies associated with lupus nephritis. J Immunol. 1989;143:103–112. - PubMed
-
- Takeuchi T, Abe T, Koide J, Hosono O, Morimoto C, Homma M. Cellular mechanism of DNA-specific antibody synthesis by lymphocytes from systemic lupus erythematosus patients. Arthritis Rheum. 1984;27:766–773. - PubMed
-
- Sieling PA, Porcelli SA, Duong BT, Spada F, Bloom BR, Diamond B, Hahn BH. Human double-negative T cells in systemic lupus erythematosus provide help for IgG and are restricted by CD1c. J Immunol. 2000;165:5338–5344. - PubMed
-
- Anand A, Dean GS, Quereshi K, Isenberg DA, Lydyard PM. Characterization of CD3+ CD4− CD8− (double negative) T cells in patients with systemic lupus erythematosus: activation markers. Lupus. 2002;11:493–500. - PubMed
-
- Rajagopalan S, Zordan T, Tsokos GC, Datta SK. Pathogenic anti-DNA autoantibody-inducing T helper cell lines from patients with active lupus nephritis: isolation of CD4-8- T helper cell lines that express the gamma delta T-cell antigen receptor. Proc Natl Acad Sci U S A. 1990;87:7020–7024. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

