Respiratory chain deficiency in aged spinal motor neurons
- PMID: 24684792
- PMCID: PMC4099519
- DOI: 10.1016/j.neurobiolaging.2014.02.027
Respiratory chain deficiency in aged spinal motor neurons
Abstract
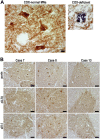
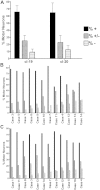
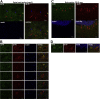
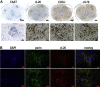
Sarcopenia, muscle wasting, and strength decline with age, is an important cause of loss of mobility in the elderly individuals. The underlying mechanisms are uncertain but likely to involve defects of motor nerve, neuromuscular junction, and muscle. Loss of motor neurons with age and subsequent denervation of skeletal muscle has been recognized as one of the contributing factors. This study investigated aspects of mitochondrial biology in spinal motor neurons from elderly subjects. We found that protein components of complex I of mitochondrial respiratory chain were reduced or absent in a proportion of aged motor neurons-a phenomenon not observed in fetal tissue. Further investigation showed that complex I-deficient cells had reduced mitochondrial DNA content and smaller soma size. We propose that mitochondrial dysfunction in these motor neurons could lead to the cell loss and ultimately denervation of muscle fibers.
Keywords: Complex I; Mitochondria; Motor neuron; Sarcopenia; Spinal cord; mtDNA.
Copyright © 2014 The Authors. Published by Elsevier Inc. All rights reserved.
Figures



References
-
- Aagaard P., Suetta C. Role of the nervous system in sarcopenia and muscle atrophy with aging: strength training as a countermeasure. Scand. J. Med. Sci. Sports. 2010;20:49–64. - PubMed
-
- Aiken J., Bua E. Mitochondrial DNA deletion mutations and sarcopenia. Ann. N. Y Acad. Sci. 2002;959:412–423. - PubMed
-
- Andersen J.L. Muscle fibre type adaptation in the elderly human muscle. Scand. J. Med. Sci. Sports. 2003;13:40–47. - PubMed
-
- Borthwick G.M., Johnson M.A. Mitochondrial enzyme activity in amyotrophic lateral sclerosis: implications for the role of mitochondria in neuronal cell death. Ann. Neurol. 1999;46:787–790. - PubMed
-
- Brown W.F., Strong M.J. Methods for estimating numbers of motor units in biceps-brachialis muscles and losses of motor units with aging. Muscle Nerve. 1988;11:423–432. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
- G000608-1/MRC_/Medical Research Council/United Kingdom
- G1100540/MRC_/Medical Research Council/United Kingdom
- 096919/Z/11/Z/WT_/Wellcome Trust/United Kingdom
- MC_PC_15004/MRC_/Medical Research Council/United Kingdom
- GR082557/WT_/Wellcome Trust/United Kingdom
- G0502157/MRC_/Medical Research Council/United Kingdom
- 096919/WT_/Wellcome Trust/United Kingdom
- G0700089/MRC_/Medical Research Council/United Kingdom
- G0400074/MRC_/Medical Research Council/United Kingdom
- G0900652/MRC_/Medical Research Council/United Kingdom
- MR/K006312/1/MRC_/Medical Research Council/United Kingdom
- G0700718/MRC_/Medical Research Council/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

