JAK/STAT pathway dysregulation in tumors: a Drosophila perspective
- PMID: 24685611
- PMCID: PMC4037387
- DOI: 10.1016/j.semcdb.2014.03.023
JAK/STAT pathway dysregulation in tumors: a Drosophila perspective
Abstract
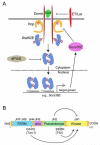
Sustained activation of the JAK/STAT pathway is causal to human cancers. This pathway is less complex in Drosophila, and its dysregulation has been linked to several tumor models in this organism. Here, we discuss models of metastatic epithelial and hematopoietic tumors that are causally linked to dysregulation of JAK/STAT signaling in Drosophila. First, we focus on cancer models in imaginal discs where ectopic expression of the JAK/STAT pathway ligand Unpaired downstream of distinct tumor suppressors has emerged as an unexpected mediator of neoplastic transformation. We also discuss the collaboration between STAT and oncogenic Ras in epithelial transformation. Second, we examine hematopoietic tumors, where mutations that cause hyperactive JAK/STAT signaling are necessary and sufficient for "fly leukemia". We highlight the important contributions that genetic screens in Drosophila have made to understanding the JAK/STAT pathway, its developmental roles, and how its function is co-opted during tumorigenesis.
Keywords: Carcinoma; Chinmo; Dome; ESCRT; Hop; Imaginal discs; JAK/STAT; Melanotic tumors; Myeloproliferative neoplasms; Notch; PRC1; Ras; Scribbled; Socs36E; Stat92E; T42; Tum-l; Upd; dPIAS.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures
Similar articles
-
Enhancer of Polycomb and the Tip60 complex repress hematological tumor initiation by negatively regulating JAK/STAT pathway activity.Dis Model Mech. 2019 May 30;12(5):dmm038679. doi: 10.1242/dmm.038679. Dis Model Mech. 2019. PMID: 31072879 Free PMC article.
-
GFP reporters detect the activation of the Drosophila JAK/STAT pathway in vivo.Gene Expr Patterns. 2007 Jan;7(3):323-31. doi: 10.1016/j.modgep.2006.08.003. Epub 2006 Aug 22. Gene Expr Patterns. 2007. PMID: 17008134
-
A Genetic Screen Reveals an Unexpected Role for Yorkie Signaling in JAK/STAT-Dependent Hematopoietic Malignancies in Drosophila melanogaster.G3 (Bethesda). 2017 Aug 7;7(8):2427-2438. doi: 10.1534/g3.117.044172. G3 (Bethesda). 2017. PMID: 28620086 Free PMC article.
-
Organogenesis and tumorigenesis: insight from the JAK/STAT pathway in the Drosophila eye.Dev Dyn. 2010 Oct;239(10):2522-33. doi: 10.1002/dvdy.22394. Dev Dyn. 2010. PMID: 20737505 Free PMC article. Review.
-
Drosophila Jak/STAT Signaling: Regulation and Relevance in Human Cancer and Metastasis.Int J Mol Sci. 2018 Dec 14;19(12):4056. doi: 10.3390/ijms19124056. Int J Mol Sci. 2018. PMID: 30558204 Free PMC article. Review.
Cited by
-
The Air Sac Primordium of Drosophila: A Model for Invasive Development.Int J Mol Sci. 2018 Jul 17;19(7):2074. doi: 10.3390/ijms19072074. Int J Mol Sci. 2018. PMID: 30018198 Free PMC article. Review.
-
Cancer cachexia: lessons from Drosophila.Dis Model Mech. 2022 Mar 1;15(3):dmm049298. doi: 10.1242/dmm.049298. Epub 2022 Mar 23. Dis Model Mech. 2022. PMID: 35319749 Free PMC article. Review.
-
SOCS-JAK-STAT inhibitors and SOCS mimetics as treatment options for autoimmune uveitis, psoriasis, lupus, and autoimmune encephalitis.Front Immunol. 2023 Oct 26;14:1271102. doi: 10.3389/fimmu.2023.1271102. eCollection 2023. Front Immunol. 2023. PMID: 38022642 Free PMC article. Review.
-
A Flippase-Mediated GAL80/GAL4 Intersectional Resource for Dissecting Appendage Development in Drosophila.G3 (Bethesda). 2015 Aug 13;5(10):2105-12. doi: 10.1534/g3.115.019810. G3 (Bethesda). 2015. PMID: 26276385 Free PMC article.
-
Discovery and evaluation of ZT55, a novel highly-selective tyrosine kinase inhibitor of JAK2V617F against myeloproliferative neoplasms.J Exp Clin Cancer Res. 2019 Feb 4;38(1):49. doi: 10.1186/s13046-019-1062-x. J Exp Clin Cancer Res. 2019. PMID: 30717771 Free PMC article.
References
-
- Abdel-Wahab O. Genetics of the myeloproliferative neoplasms. Curr Opin Hematol. 2011;18(2):117–23. - PubMed
-
- Arbouzova NI, Bach EA, Zeidler MP. Ken & barbie selectively regulates the expression of a subset of Jak/STAT pathway target genes. Curr Biol. 2006;16(1):80–8. - PubMed
-
- Arbouzova NI, Zeidler MP. JAK/STAT signalling in Drosophila: insights into conserved regulatory and cellular functions. Development. 2006;133(14):2605–16. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

