The Mark Coventry Award: Trabecular metal tibial components were durable and reliable in primary total knee arthroplasty: a randomized clinical trial
- PMID: 24687433
- PMCID: PMC4390908
- DOI: 10.1007/s11999-014-3585-y
The Mark Coventry Award: Trabecular metal tibial components were durable and reliable in primary total knee arthroplasty: a randomized clinical trial
Abstract
Background: Although highly porous metals have demonstrated excellent bone ingrowth properties and so are an intriguing option for fixation in total knee arthroplasty (TKA), some surgeons are skeptical about the durability of uncemented tibial fixation and the potential for soft tissues to adhere to these porous metals and perhaps cause knee stiffness or pain.
Questions/purposes: The purpose of this study was to compare, in the context of a randomized clinical trial, a highly porous metal tibia compared with a traditional modular cemented tibia in terms of survivorship, Knee Society scores, range of motion (ROM), and complications.
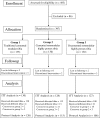
Methods: From 2003 to 2006, 397 patients (age 67.8 ± 8.7 years; 54% female) were randomized to three groups: (1) traditional modular cemented tibia; (2) cemented highly porous metal tibia; and (3) uncemented highly porous metal tibia. The same posterior-stabilized femoral component and patella component were cemented in every case. Stratified randomization was done for surgeon, patient's age, sex, and body mass index. Survivorship at 5 years was compared between the groups, as were Knee Society scores, ROM, and complications. Radiographic assessment included alignment, radiolucency, and implant migration/loosening. Patients were followed until death, revision, or for a minimum of 2 years (mean, 5 years; range, 2-9 years). Four patients were lost to followup before 2 years.
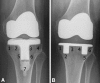
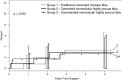
Results: Highly porous metal tibias (both uncemented and cemented) were no different from traditional cemented modular tibial modular components in terms of survivorship at 5 years using a intention-to-treat analysis (96.8% [1]; 97.6% [2]; 96.7% [3]; p = 0.59). A per-protocol analysis revealed that no highly porous metal tibia was revised for aseptic loosening. Highly porous metal tibias performed comparably to traditional cemented modular tibias in terms of Knee Society scores, ROM, and the frequency of complications.
Conclusions: At 5 years this randomized clinical trial demonstrated that highly porous metal tibias provided comparably durable fixation and reliable pain relief and restoration of function when compared with a traditional cemented modular tibia in TKA.
Level of evidence: Level I, therapeutic study. See Guidelines for Authors for a complete description of levels of evidence.
Figures

Comment in
-
CORR Insights®: the Mark Coventry Award: Trabecular metal tibial components were durable and reliable in primary total knee arthroplasty: a randomized clinical trial.Clin Orthop Relat Res. 2015 Jan;473(1):43-4. doi: 10.1007/s11999-014-3656-0. Epub 2014 Apr 30. Clin Orthop Relat Res. 2015. PMID: 24781927 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

