Probing Genome Maintenance Functions of human RECQ1
- PMID: 24688722
- PMCID: PMC3962141
- DOI: 10.5936/csbj.201303014
Probing Genome Maintenance Functions of human RECQ1
Abstract
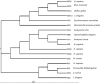
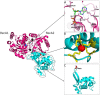
The RecQ helicases are a highly conserved family of DNA-unwinding enzymes that play key roles in protecting the genome stability in all kingdoms of life. Human RecQ homologs include RECQ1, BLM, WRN, RECQ4, and RECQ5β. Although the individual RecQ-related diseases are characterized by a variety of clinical features encompassing growth defects (Bloom Syndrome and Rothmund Thomson Syndrome) to premature aging (Werner Syndrome), all these patients have a high risk of cancer predisposition. Here, we present an overview of recent progress towards elucidating functions of RECQ1 helicase, the most abundant but poorly characterized RecQ homolog in humans. Consistent with a conserved role in genome stability maintenance, deficiency of RECQ1 results in elevated frequency of spontaneous sister chromatid exchanges, chromosomal instability, increased DNA damage and greater sensitivity to certain genotoxic stress. Delineating what aspects of RECQ1 catalytic functions contribute to the observed cellular phenotypes, and how this is regulated is critical to establish its biological functions in DNA metabolism. Recent studies have identified functional specialization of RECQ1 in DNA repair; however, identification of fundamental similarities will be just as critical in developing a unifying theme for RecQ actions, allowing the functions revealed from studying one homolog to be extrapolated and generalized to other RecQ homologs.
Keywords: DNA damage; DNA repair; Helicase; RecQ; genomic instability.
Figures

References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
