Targeting TGF-β Signaling by Antisense Oligonucleotide-mediated Knockdown of TGF-β Type I Receptor
- PMID: 24691207
- PMCID: PMC4011125
- DOI: 10.1038/mtna.2014.7
Targeting TGF-β Signaling by Antisense Oligonucleotide-mediated Knockdown of TGF-β Type I Receptor
Abstract
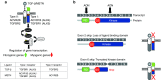
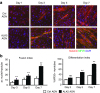
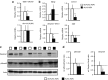
Duchenne muscular dystrophy (DMD) is caused by lack of functional dystrophin and results in progressive myofiber damage and degeneration. In addition, impaired muscle regeneration and fibrosis contribute to the progressive pathology of DMD. Importantly, transforming growth factor-β (TGF-β) is implicated in DMD pathology and is known to stimulate fibrosis and inhibit muscle regeneration. In this study, we present a new strategy to target TGF-β signaling cascades by specifically inhibiting the expression of TGF-β type I receptor TGFBR1 (ALK5). Antisense oligonucleotides (AONs) were designed to specifically induce exon skipping of mouse ALK5 transcripts. AON-induced exon skipping of ALK5 resulted in specific downregulation of full-length receptor transcripts in vitro in different cell types, repression of TGF-β activity, and enhanced C2C12 myoblast differentiation. To determine the effect of these AONs in dystrophic muscles, we performed intramuscular injections of ALK5 AONs in mdx mice, which resulted in a decrease in expression of fibrosis-related genes and upregulation of Myog expression compared to control AON-injected muscles. In summary, our study presents a novel method to target TGF-β signaling cascades with potential beneficial effects for DMD.
Figures


Similar articles
-
Dual exon skipping in myostatin and dystrophin for Duchenne muscular dystrophy.BMC Med Genomics. 2011 Apr 20;4:36. doi: 10.1186/1755-8794-4-36. BMC Med Genomics. 2011. PMID: 21507246 Free PMC article.
-
Exon 51 Skipping Quantification by Digital Droplet PCR in del52hDMD/mdx Mice.Methods Mol Biol. 2018;1828:249-262. doi: 10.1007/978-1-4939-8651-4_15. Methods Mol Biol. 2018. PMID: 30171546
-
In vivo comparison of 2'-O-methyl phosphorothioate and morpholino antisense oligonucleotides for Duchenne muscular dystrophy exon skipping.J Gene Med. 2009 Mar;11(3):257-66. doi: 10.1002/jgm.1288. J Gene Med. 2009. PMID: 19140108
-
Optimization of antisense-mediated exon skipping for Duchenne muscular dystrophy.Gene Ther. 2020 Sep;27(9):407-416. doi: 10.1038/s41434-020-0156-6. Epub 2020 Jun 1. Gene Ther. 2020. PMID: 32483212 Review.
-
Molecular correction of Duchenne muscular dystrophy by splice modulation and gene editing.RNA Biol. 2021 Jul;18(7):1048-1062. doi: 10.1080/15476286.2021.1874161. Epub 2021 Jan 20. RNA Biol. 2021. PMID: 33472516 Free PMC article. Review.
Cited by
-
New function of the myostatin/activin type I receptor (ALK4) as a mediator of muscle atrophy and muscle regeneration.FASEB J. 2017 Jan;31(1):238-255. doi: 10.1096/fj.201600675R. Epub 2016 Oct 12. FASEB J. 2017. PMID: 27733450 Free PMC article.
-
Regenerative inflammation: When immune cells help to re-build tissues.FEBS J. 2024 Apr;291(8):1597-1614. doi: 10.1111/febs.16693. Epub 2022 Dec 15. FEBS J. 2024. PMID: 36440547 Free PMC article. Review.
-
Recent Advancements in Antifibrotic Therapies for Regression of Liver Fibrosis.Cells. 2022 Apr 29;11(9):1500. doi: 10.3390/cells11091500. Cells. 2022. PMID: 35563807 Free PMC article. Review.
-
Role of Transforming Growth Factor-β in Skeletal Muscle Fibrosis: A Review.Int J Mol Sci. 2019 May 17;20(10):2446. doi: 10.3390/ijms20102446. Int J Mol Sci. 2019. PMID: 31108916 Free PMC article. Review.
-
Current Pharmacological Strategies for Duchenne Muscular Dystrophy.Front Cell Dev Biol. 2021 Aug 19;9:689533. doi: 10.3389/fcell.2021.689533. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34490244 Free PMC article. Review.
References
-
- Bushby K, Finkel R, Birnkrant DJ, Case LE, Clemens PR, Cripe L, et al. DMD Care Considerations Working Group Diagnosis and management of Duchenne muscular dystrophy, part 1: diagnosis, and pharmacological and psychosocial management. Lancet Neurol. 2010;9:77–93. - PubMed
-
- Blake DJ, Weir A, Newey SE, Davies KE. Function and genetics of dystrophin and dystrophin-related proteins in muscle. Physiol Rev. 2002;82:291–329. - PubMed
-
- Bushby K, Finkel R, Birnkrant DJ, Case LE, Clemens PR, Cripe L, et al. DMD Care Considerations Working Group Diagnosis and management of Duchenne muscular dystrophy, part 2: implementation of multidisciplinary care. Lancet Neurol. 2010;9:177–189. - PubMed
-
- Cirak S, Arechavala-Gomeza V, Guglieri M, Feng L, Torelli S, Anthony K, et al. Exon skipping and dystrophin restoration in patients with Duchenne muscular dystrophy after systemic phosphorodiamidate morpholino oligomer treatment: an open-label, phase 2, dose-escalation study. Lancet. 2011;378:595–605. - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

