Effect of protein binding on unbound atazanavir and darunavir cerebrospinal fluid concentrations
- PMID: 24691856
- PMCID: PMC4396178
- DOI: 10.1002/jcph.298
Effect of protein binding on unbound atazanavir and darunavir cerebrospinal fluid concentrations
Abstract
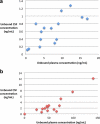
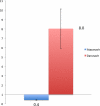
HIV-1 protease inhibitors (PIs) exhibit different protein binding affinities and achieve variable plasma and tissue concentrations. Degree of plasma protein binding may impact central nervous system penetration. This cross-sectional study assessed cerebrospinal fluid (CSF) unbound PI concentrations, HIV-1 RNA, and neopterin levels in subjects receiving either ritonavir-boosted darunavir (DRV), 95% plasma protein bound, or atazanavir (ATV), 86% bound. Unbound PI trough concentrations were measured using rapid equilibrium dialysis and liquid chromatography/tandem mass spectrometry. Plasma and CSF HIV-1 RNA and neopterin were measured by Ampliprep/COBAS® Taqman® 2.0 assay (Roche) and enzyme-linked immunosorbent assay (ALPCO), respectively. CSF/plasma unbound drug concentration ratio was higher for ATV, 0.09 [95% confidence interval (CI) 0.06-0.12] than DRV, 0.04 (95%CI 0.03-0.06). Unbound CSF concentrations were lower than protein adjusted wild-type inhibitory concentration-50 (IC50 ) in all ATV and 1 DRV-treated subjects (P < 0.001). CSF HIV-1 RNA was detected in 2/15 ATV and 4/15 DRV subjects (P = 0.65). CSF neopterin levels were low and similar between arms. ATV relative to DRV had higher CSF/plasma unbound drug ratio. Low CSF HIV-1 RNA and neopterin suggest that both regimens resulted in CSF virologic suppression and controlled inflammation.
Keywords: HIV/AIDS; central nervous system; clinical pharmacology.
© 2014, The American College of Clinical Pharmacology.
Figures
Similar articles
-
Ritonavir-boosted darunavir is rarely associated with nephrolithiasis compared with ritonavir-boosted atazanavir in HIV-infected patients.PLoS One. 2013 Oct 10;8(10):e77268. doi: 10.1371/journal.pone.0077268. eCollection 2013. PLoS One. 2013. PMID: 24130871 Free PMC article.
-
Metabolic effects of darunavir/ritonavir versus atazanavir/ritonavir in treatment-naive, HIV type 1-infected subjects over 48 weeks.AIDS Res Hum Retroviruses. 2012 Oct;28(10):1184-95. doi: 10.1089/aid.2011.0327. Epub 2012 Apr 2. AIDS Res Hum Retroviruses. 2012. PMID: 22352336 Free PMC article. Clinical Trial.
-
Cerebrospinal fluid drug concentrations and viral suppression in HIV-1-infected patients receiving ritonavir-boosted atazanavir plus lamivudine dual antiretroviral therapy (Spanish HIV/AIDS Research Network, PreEC/RIS 39).J Neurovirol. 2018 Aug;24(4):391-397. doi: 10.1007/s13365-018-0626-4. Epub 2018 Mar 14. J Neurovirol. 2018. PMID: 29542028
-
Cobicistat: a review of its use as a pharmacokinetic enhancer of atazanavir and darunavir in patients with HIV-1 infection.Drugs. 2014 Feb;74(2):195-206. doi: 10.1007/s40265-013-0160-x. Drugs. 2014. PMID: 24343782 Review.
-
Clinical pharmacokinetics and summary of efficacy and tolerability of atazanavir.Clin Pharmacokinet. 2005;44(10):1035-50. doi: 10.2165/00003088-200544100-00003. Clin Pharmacokinet. 2005. PMID: 16176117 Review.
Cited by
-
Central nervous system disorders after use of dolutegravir: evidence from preclinical and clinical studies.Pharmacol Rep. 2023 Oct;75(5):1138-1151. doi: 10.1007/s43440-023-00515-y. Epub 2023 Aug 21. Pharmacol Rep. 2023. PMID: 37605102 Free PMC article. Review.
-
Evaluation of cerebrospinal fluid virological escape in patients on long-term protease inhibitor monotherapy.Antivir Ther. 2017;22(6):535-538. doi: 10.3851/IMP3146. Epub 2017 Feb 24. Antivir Ther. 2017. PMID: 28234235 Free PMC article.
-
Pharmacokinetics and pharmacodynamics of antiretrovirals in the central nervous system.Clin Pharmacokinet. 2014 Oct;53(10):891-906. doi: 10.1007/s40262-014-0171-0. Clin Pharmacokinet. 2014. PMID: 25200312 Review.
-
Impact of protease inhibitors on intracellular concentration of tenofovir-diphosphate among HIV-1 infected patients.AIDS. 2015 Jun 1;29(9):1113-5. doi: 10.1097/QAD.0000000000000659. AIDS. 2015. PMID: 25870991 Free PMC article.
-
Pharmacologic approaches to HIV-associated neurocognitive disorders.Curr Opin Pharmacol. 2020 Oct;54:102-108. doi: 10.1016/j.coph.2020.09.003. Epub 2020 Oct 10. Curr Opin Pharmacol. 2020. PMID: 33049585 Free PMC article. Review.
References
-
- Palmer S, Josefsson L, Coffin JM. HIV reservoirs and the possibility of a cure for HIV infection. J Intern Med. 2011;270(6):550–560. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

