Multiphasic construct studied in an ectopic osteochondral defect model
- PMID: 24694896
- PMCID: PMC4006259
- DOI: 10.1098/rsif.2014.0184
Multiphasic construct studied in an ectopic osteochondral defect model
Abstract
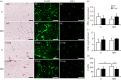
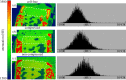
In vivo osteochondral defect models predominantly consist of small animals, such as rabbits. Although they have an advantage of low cost and manageability, their joints are smaller and more easily healed compared with larger animals or humans. We hypothesized that osteochondral cores from large animals can be implanted subcutaneously in rats to create an ectopic osteochondral defect model for routine and high-throughput screening of multiphasic scaffold designs and/or tissue-engineered constructs (TECs). Bovine osteochondral plugs with 4 mm diameter osteochondral defect were fitted with novel multiphasic osteochondral grafts composed of chondrocyte-seeded alginate gels and osteoblast-seeded polycaprolactone scaffolds, prior to being implanted in rats subcutaneously with bone morphogenic protein-7. After 12 weeks of in vivo implantation, histological and micro-computed tomography analyses demonstrated that TECs are susceptible to mineralization. Additionally, there was limited bone formation in the scaffold. These results suggest that the current model requires optimization to facilitate robust bone regeneration and vascular infiltration into the defect site. Taken together, this study provides a proof-of-concept for a high-throughput osteochondral defect model. With further optimization, the presented hybrid in vivo model may address the growing need for a cost-effective way to screen osteochondral repair strategies before moving to large animal preclinical trials.
Keywords: biomaterials; in vivo model; multiphasic scaffold; osteochondral repair; tissue engineering.
Figures






References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
