Discovery of a neuroprotective chemical, (S)-N-(3-(3,6-dibromo-9H-carbazol-9-yl)-2-fluoropropyl)-6-methoxypyridin-2-amine [(-)-P7C3-S243], with improved druglike properties
- PMID: 24697290
- PMCID: PMC4206694
- DOI: 10.1021/jm401919s
Discovery of a neuroprotective chemical, (S)-N-(3-(3,6-dibromo-9H-carbazol-9-yl)-2-fluoropropyl)-6-methoxypyridin-2-amine [(-)-P7C3-S243], with improved druglike properties
Abstract
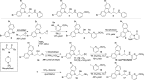
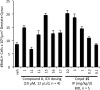
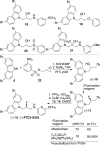
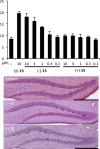
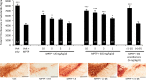
(-)-P7C3-S243 is a neuroprotective aminopropyl carbazole with improved druglike properties compared with previously reported compounds in the P7C3 class. It protects developing neurons in a mouse model of hippocampal neurogenesis and protects mature neurons within the substantia nigra in a mouse model of Parkinson's disease. A short, enantioselective synthesis provides the neuroprotective agent in optically pure form. It is nontoxic, orally bioavailable, metabolically stable, and able to cross the blood-brain barrier. As such, it represents a valuable lead compound for the development of drugs to treat neurodegenerative diseases and traumatic brain injury.
Figures
References
-
- Pieper A. A.; Xie S.; Capota E.; Estill S. J.; Zhong J.; Long J. M.; Becker G. L.; Huntington P.; Goldman S. E.; Shen C.-H.; Capota M.; Britt J. K.; Kotti T.; Ure K.; Brat D. J.; Williams N. S.; MacMillan K. S.; Naidoo J.; Melito L.; Hsieh J.; De Brabander J.; Ready J. M.; McKnight S. L. Discovery of a Proneurogenic, Neuroprotective Chemical. Cell 2010, 142, 39–51. - PMC - PubMed
- McKnight S. L.; Pieper A. A.; Ready J. M.; De Brabander J.. Proneurogenic Compounds. U.S. Patent 8,604,074, 2013.
- McKnight S. L.; Pieper A. A.; Ready J. M.; De Brabander J.. Proneurogenic Compounds. U.S. Patent 8,362,277, 2013.
-
- MacMillan K. S.; Naidoo J.; Liang J.; Melito L.; Williams N. S.; Morlock L.; Huntington P. J.; Estill S. J.; Longgood J.; Becker G. L.; McKnight S. L.; Pieper A. A.; De Brabander J. K.; Ready J. M. Development of Proneurogenic, Neuroprotective Small Molecules. J. Am. Chem. Soc. 2011, 133, 1428–1437. - PMC - PubMed
-
- Naidoo J.; Bemben C. J.; Allwein S. R.; Liang J.; Pieper A. A.; Ready J. M. Development of a Scalable Synthesis of P7C3-A20, a Potent Neuroprotective Agent. Tetrahedron Lett. 2013, 54, 4429–4431.
-
- De Jesus-Cortes H.; Xu P.; Drawbridge J.; Estill S. J.; Huntington P.; Tran S.; Britt J.; Tesla R.; Morlock L.; Naidoo J.; Melito L. M.; Wang G.; Williams N. S.; Ready J. M.; McKnight S. L.; Pieper A. A. Neuroprotective Efficacy of Aminopropyl Carbazoles in a Mouse Model of Parkinson Disease. Proc. Natl. Acad. Sci. U.S.A. 2012, 109, 17010–17015. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Research Materials

