Longitudinally extensive NMO spinal cord pathology produced by passive transfer of NMO-IgG in mice lacking complement inhibitor CD59
- PMID: 24698947
- PMCID: PMC4162764
- DOI: 10.1016/j.jaut.2014.02.011
Longitudinally extensive NMO spinal cord pathology produced by passive transfer of NMO-IgG in mice lacking complement inhibitor CD59
Abstract
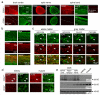
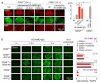
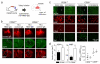
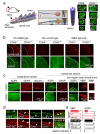
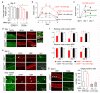
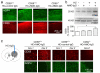
Spinal cord pathology with inflammatory, demyelinating lesions spanning three or more vertebral segments is a characteristic feature of neuromyelitis optica (NMO). NMO pathogenesis is thought to involve binding of immunoglobulin G anti-aquaporin-4 autoantibodies (NMO-IgG) to astrocytes, causing complement-dependent cytotoxicity (CDC) and secondary inflammation, demyelination and neuron loss. We investigated the involvement of CD59, a glycophosphoinositol (GPI)-anchored membrane protein on astrocytes that inhibits formation of the terminal C5b-9 membrane attack complex. CD59 inhibition by a neutralizing monoclonal antibody greatly increased NMO-IgG-dependent CDC in murine astrocyte cultures and ex vivo spinal cord slice cultures. Greatly increased NMO pathology was also found in spinal cord slice cultures from CD59 knockout mice, and in vivo following intracerebral injection of NMO-IgG and human complement. Intrathecal injection (at L5-L6) of small amounts of NMO-IgG and human complement in CD59-deficient mice produced robust, longitudinally extensive white matter lesions in lumbar spinal cord. Pathology was most severe at day 2 after injection, showing loss of AQP4 and GFAP, C5b-9 deposition, microglial activation, granulocyte infiltration, and demyelination. Hind limb motor function was remarkably impaired as well. There was partial remyelination and recovery of motor function by day 5. Our results implicate CD59 as an important modulator of the immune response in NMO, and provide a novel animal model of NMO that closely recapitulates human NMO pathology. Up-regulation of CD59 on astrocytes may have therapeutic benefit in NMO.
Keywords: Aquaporin-4; Astrocyte; Complement-dependent cytotoxicity; Demyelination; NMO.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures

References
-
- Jarius S, Paul F, Franciotta D, Waters P, Zipp F, Hohlfeld R, et al. Mechanisms of disease: aquaporin-4 antibodies in neuromyelitis optica. Nat Clin Pract Neurol. 2008;4:202–14. - PubMed
-
- Wingerchuk DM, Lennon VA, Lucchinetti CF, Pittock SJ, Weinshenker BG. The spectrum of neuromyelitis optica. Lancet Neurol. 2007;6:805–15. - PubMed
-
- Jarius S, Wildemann B. AQP4 antibodies in neuromyelitis optica: diagnostic and pathogenetic relevance. Nat Rev Neurol. 2010;6:383–92. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

