Doppler optical coherence tomography
- PMID: 24704352
- PMCID: PMC4073226
- DOI: 10.1016/j.preteyeres.2014.03.004
Doppler optical coherence tomography
Abstract
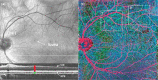
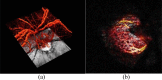
Optical Coherence Tomography (OCT) has revolutionized ophthalmology. Since its introduction in the early 1990s it has continuously improved in terms of speed, resolution and sensitivity. The technique has also seen a variety of extensions aiming to assess functional aspects of the tissue in addition to morphology. One of these approaches is Doppler OCT (DOCT), which aims to visualize and quantify blood flow. Such extensions were already implemented in time domain systems, but have gained importance with the introduction of Fourier domain OCT. Nowadays phase-sensitive detection techniques are most widely used to extract blood velocity and blood flow from tissues. A common problem with the technique is that the Doppler angle is not known and several approaches have been realized to obtain absolute velocity and flow data from the retina. Additional studies are required to elucidate which of these techniques is most promising. In the recent years, however, several groups have shown that data can be obtained with high validity and reproducibility. In addition, several groups have published values for total retinal blood flow. Another promising application relates to non-invasive angiography. As compared to standard techniques such as fluorescein and indocyanine-green angiography the technique offers two major advantages: no dye is required and depth resolution is required is provided. As such Doppler OCT has the potential to improve our abilities to diagnose and monitor ocular vascular diseases.
Keywords: Angiography; Blood flow; Doppler effect; Optical Coherence Tomography; Perfusion; Retinal vasculature.
Copyright © 2014 The Authors. Published by Elsevier Ltd.. All rights reserved.
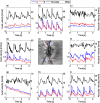
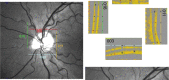
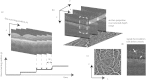
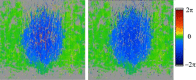
Figures





References
-
- Ahmad Fadzil M.H., Izhar L.I., Nugroho H.A. Determination of foveal avascular zone in diabetic retinopathy digital fundus images. Comput. Biol. Med. 2010;40:657–664. - PubMed
-
- Alipour S.H., Rabbani H., Akhlaghi M., Dehnavi A.M., Javanmard S.H. Analysis of foveal avascular zone for grading of diabetic retinopathy severity based on curvelet transform. Graefes Arch. Clin. Exp. Ophthalmol. 2012;250:1607–1614. - PubMed
-
- An L., Wang R.K. In vivo volumetric imaging of vascular perfusion within human retina and choroids with optical micro-angiography. Opt. Express. 2008;16:11438–11452. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

