Opposing microtubule motors control motility, morphology and cargo segregation during ER-to-Golgi transport
- PMID: 24705013
- PMCID: PMC4021352
- DOI: 10.1242/bio.20147633
Opposing microtubule motors control motility, morphology and cargo segregation during ER-to-Golgi transport
Abstract
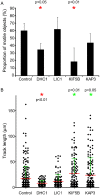
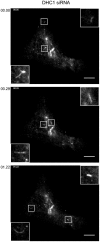
We recently demonstrated that dynein and kinesin motors drive multiple aspects of endosomal function in mammalian cells. These functions include driving motility, maintaining morphology (notably through providing longitudinal tension to support vesicle fission), and driving cargo sorting. Microtubule motors drive bidirectional motility during traffic between the endoplasmic reticulum (ER) and Golgi. Here, we have examined the role of microtubule motors in transport carrier motility, morphology, and domain organization during ER-to-Golgi transport. We show that, consistent with our findings for endosomal dynamics, microtubule motor function during ER-to-Golgi transport of secretory cargo is required for motility, morphology, and cargo sorting within vesicular tubular carriers en route to the Golgi. Our data are consistent with previous findings that defined roles for dynein-1, kinesin-1 (KIF5B) and kinesin-2 in this trafficking step. Our high resolution tracking data identify some intriguing aspects. Depletion of kinesin-1 reduces the number of motile structures seen, which is in line with other findings relating to the role of kinesin-1 in ER export. However, those transport carriers that were produced had a much greater run length suggesting that this motor can act as a brake on anterograde motility. Kinesin-2 depletion did not significantly reduce the number of motile transport carriers but did cause a similar increase in run length. These data suggest that kinesins act as negative regulators of ER-to-Golgi transport. Depletion of dynein not only reduced the number of motile carriers formed but also caused tubulation of carriers similar to that seen for sorting nexin-coated early endosomes. Our data indicated that the previously observed anterograde-retrograde polarity of transport carriers in transit to the Golgi from the ER is maintained by microtubule motor function.
Keywords: Dynein; Endoplasmic reticulum; Golgi; Kinesin; Microtubule motor; Secretory cargo trafficking.
© 2014. Published by The Company of Biologists Ltd.
Conflict of interest statement
Figures

References
-
- Chen C. A., Okayama H. (1988). Calcium phosphate-mediated gene transfer: a highly efficient transfection system for stably transforming cells with plasmid DNA. Biotechniques 6, 632–638. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

