Assessment of fecundity and germ line transmission in two transgenic pig lines produced by sleeping beauty transposition
- PMID: 24705079
- PMCID: PMC3899982
- DOI: 10.3390/genes3040615
Assessment of fecundity and germ line transmission in two transgenic pig lines produced by sleeping beauty transposition
Abstract
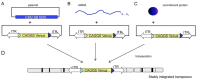
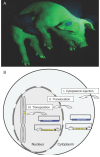
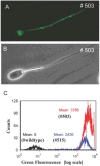
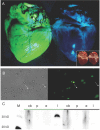
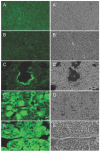
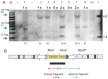
Recently, we described a simplified injection method for producing transgenic pigs using a non-autonomous Sleeping Beauty transposon system. The founder animals showed ubiquitous expression of the Venus fluorophore in almost all cell types. To assess, whether expression of the reporter fluorophore affects animal welfare or fecundity, we analyzed reproductive parameters of two founder boars, germ line transmission, and organ and cell specific transgene expression in animals of the F1 and F2 generation. Molecular analysis of ejaculated sperm cells suggested three monomeric integrations of the Venus transposon in both founders. To test germ line transmission of the three monomeric transposon integrations, wild-type sows were artificially inseminated. The offspring were nursed to sexual maturity and hemizygous lines were established. A clear segregation of the monomeric transposons following the Mendelian rules was observed in the F1 and F2 offspring. Apparently, almost all somatic cells, as well as oocytes and spermatozoa, expressed the Venus fluorophore at cell-type specific levels. No detrimental effects of Venus expression on animal health or fecundity were found. Importantly, all hemizygous lines expressed the fluorophore in comparable levels, and no case of transgene silencing or variegated expression was found after germ line transmission, suggesting that the insertions occurred at transcriptionally permissive loci. The results show that Sleeping Beauty transposase-catalyzed transposition is a promising approach for stable genetic modification of the pig genome.
Figures

Similar articles
-
Germline transgenic pigs by Sleeping Beauty transposition in porcine zygotes and targeted integration in the pig genome.PLoS One. 2011;6(8):e23573. doi: 10.1371/journal.pone.0023573. Epub 2011 Aug 29. PLoS One. 2011. PMID: 21897845 Free PMC article.
-
Insertional mutagenesis by a hybrid piggyBac and sleeping beauty transposon in the rat.Genetics. 2012 Dec;192(4):1235-48. doi: 10.1534/genetics.112.140855. Epub 2012 Sep 28. Genetics. 2012. PMID: 23023007 Free PMC article.
-
Genotype-independent transmission of transgenic fluorophore protein by boar spermatozoa.PLoS One. 2011;6(11):e27563. doi: 10.1371/journal.pone.0027563. Epub 2011 Nov 16. PLoS One. 2011. PMID: 22110672 Free PMC article.
-
Sleeping beauty transposition: biology and applications for molecular therapy.Mol Ther. 2004 Feb;9(2):147-56. doi: 10.1016/j.ymthe.2003.11.009. Mol Ther. 2004. PMID: 14759798 Review.
-
Sleeping Beauty transposon system for genetic etiological research and gene therapy of cancers.Cancer Biol Ther. 2015;16(1):8-16. doi: 10.4161/15384047.2014.986944. Cancer Biol Ther. 2015. PMID: 25455252 Free PMC article. Review.
Cited by
-
Derivation and characterization of bovine induced pluripotent stem cells by transposon-mediated reprogramming.Cell Reprogram. 2015 Apr;17(2):131-40. doi: 10.1089/cell.2014.0080. Cell Reprogram. 2015. PMID: 25826726 Free PMC article.
-
Characterization of Growth and Reproduction Performance, Transgene Integration, Expression, and Transmission Patterns in Transgenic Pigs Produced by piggyBac Transposition-Mediated Gene Transfer.Anim Biotechnol. 2016 Oct;27(4):245-55. doi: 10.1080/10495398.2016.1178140. Anim Biotechnol. 2016. PMID: 27565868 Free PMC article.
-
Current progress of genetically engineered pig models for biomedical research.Biores Open Access. 2014 Dec 1;3(6):255-64. doi: 10.1089/biores.2014.0039. Biores Open Access. 2014. PMID: 25469311 Free PMC article. Review.
-
Expression of Active Fluorophore Proteins in the Milk of Transgenic Pigs Bypassing the Secretory Pathway.Sci Rep. 2016 Apr 18;6:24464. doi: 10.1038/srep24464. Sci Rep. 2016. PMID: 27086548 Free PMC article.
-
Potential of transposon-mediated cellular reprogramming towards cell-based therapies.World J Stem Cells. 2020 Jul 26;12(7):527-544. doi: 10.4252/wjsc.v12.i7.527. World J Stem Cells. 2020. PMID: 32843912 Free PMC article. Review.
References
-
- Jacobsen J.C., Bawden C.S., Rudiger S.R., McLaughlan C.J., Reid S.J., Waldvogel H.J., MacDonald M.E., Gusella J.F., Walker S.K., Kelly J.M., Webb G.C., Faull R.L., Rees M.I., Snell R.G. An ovine transgenic, Huntington's disease model. Hum. Mol. Genet. 2010;19:1873–1882. doi: 10.1093/hmg/ddq063. - DOI - PMC - PubMed
-
- Kragh P.M., Nielsen A.L., Li J., Du Y., Lin L., Schmidt M., Bogh I.B., Holm I.E., Jakobsen J.E., Johansen M.G., Purup S., Bolund L., Vajta G., Jorgensen A.L. Hemizygous minipigs produced by random gene insertion and handmade cloning express the Alzheimer's disease-causing dominant mutation APPsw. Transgenic. Res. 2009;18:545–558. doi: 10.1007/s11248-009-9245-4. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

