Expression of thyrotropin receptor, thyroglobulin, sodium-iodide symporter, and thyroperoxidase by fibrocytes depends on AIRE
- PMID: 24708100
- PMCID: PMC4079309
- DOI: 10.1210/jc.2013-4271
Expression of thyrotropin receptor, thyroglobulin, sodium-iodide symporter, and thyroperoxidase by fibrocytes depends on AIRE
Abstract
Context: CD34(+) fibrocytes, bone marrow-derived progenitor cells, infiltrate orbital connective tissue in thyroid-associated ophthalmopathy, a manifestation of Graves' disease. In the orbit, they become CD34(+) fibroblasts and coexist with native CD34(-) fibroblasts. Fibrocytes have been shown to express TSH receptor and thyroglobulin.
Objective: The objective of the study was to determine whether a broader repertoire of thyroid protein expression can be detected in fibrocytes and whether a common factor is responsible.
Design/setting/participants: Fibrocytes and fibroblasts were collected and analyzed from healthy individuals and those with Graves' disease in an academic clinical practice.
Main outcome measures: Real-time PCR, Western blot analysis, gene promoter analysis, cell transfections, and flow cytometric cell sorting were performed.
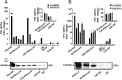
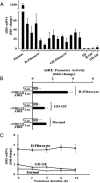
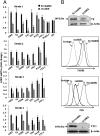
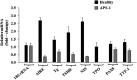
Results: We detect two additional thyroid proteins expressed by fibrocytes, namely sodium-iodide symporter and thyroperoxidase. The autoimmune regulator (AIRE) protein appears necessary for this expression. AIRE expression in fibrocytes results from an active AIRE gene promoter and stable AIRE mRNA. Knocking down AIRE with a targeting small interfering RNA reduces the expression of these thyroid proteins in fibrocytes as well as the transcription factors paired box-8 and thyroid transcription factor-1. When compared with an unaffected first-degree relative, levels of these proteins are substantially reduced in fibrocytes from an individual with an inactivating AIRE mutation. Levels of AIRE and the thyroid proteins are lower in orbital fibroblasts from patients with thyroid-associated ophthalmopathy than in fibrocytes. However, when mixed fibroblast populations are sorted into pure CD34(+) and CD34(-) subsets, the levels of these proteins are dramatically increased selectively in CD34(+) fibroblasts.
Conclusions: Fibrocytes express four proteins, the aggregate expression of which was previously thought to be restricted to thyroid epithelium. These proteins represent the necessary molecular biosynthetic machinery necessary for thyroid hormone production. Our findings implicate AIRE in the promiscuous expression of thyroid proteins in fibrocytes.
Figures

References
-
- Wang J, Jiao H, Stewart TL, Shankowsky HA, Scott PG, Tredget EE. Improvement in postburn hypertrophic scar after treatment with IFN-α2b is associated with decreased fibrocytes. J Interferon Cytokine Res. 2007;27:921–930 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

