Grass-specific CD4(+) T-cells exhibit varying degrees of cross-reactivity, implications for allergen-specific immunotherapy
- PMID: 24708411
- PMCID: PMC4069242
- DOI: 10.1111/cea.12324
Grass-specific CD4(+) T-cells exhibit varying degrees of cross-reactivity, implications for allergen-specific immunotherapy
Abstract
Background: Conceptually, allergic responses may involve cross-reactivity by antibodies or T-cells. While IgE cross-reactivity among grass-pollen allergens has been observed, cross-reactivity at the allergen-specific T-cell level has been less documented. Identification of the patterns of cross-reactivity may improve our understanding, allowing optimization of better immunotherapy strategies.
Objectives: We use Phleum pratense as model for the studying of cross-reactivity at the allergen-specific CD4(+) T cell level among DR04:01 restricted Pooideae grass-pollen T-cell epitopes.
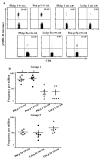
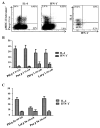
Methods: After in vitro culture of blood mono-nucleated cells from grass-pollen-allergic subjects with specific Pooideae antigenic epitopes, dual tetramer staining with APC-labelled DR04:01/Phleum pratense tetramers and PE-labelled DR04:01/Pooideae grass homolog tetramers was assessed to identify cross-reactivity among allergen-specific DR04:01-restricted T-cells in six subjects. Direct ex vivo staining enabled the comparison of frequency and phenotype of different Pooideae grass-pollen reactive T-cells. Intracellular cytokine staining (ICS) assays were also used to examine phenotypes of these T-cells.
Results: T-cells with various degrees of cross-reactive profiles could be detected. Poa p 1 97-116 , Lol p 1 221-240 , Lol p 5a 199-218 , and Poa p 5a 199-218 were identified as minimally cross-reactive T-cell epitopes that do not show cross-reactivity to Phl p 1 and Phl p 5a epitopes. Ex vivo tetramer staining assays demonstrated T-cells that recognized these minimally cross-reactive T-cell epitopes are present in Grass-pollen-allergic subjects.
Conclusions: Our results suggest that not all Pooideae grass epitopes with sequence homology are cross-reactive. Non-cross-reactive T-cells with comparable frequency, phenotype and functionality to Phl p-specific T-cells suggest that a multiple allergen system should be considered for immunotherapy instead of a mono-allergen system.
Keywords: CD4+; MHC class II tetramers; Pooideae; T-cells; allergy; cross-reactivity; epitopes; grass-pollen.
© 2014 John Wiley & Sons Ltd.
Figures




Comment in
-
The specifics of allergen recognition by CD4(+) T lymphocytes at the epitope level.Clin Exp Allergy. 2014 Jul;44(7):898-900. doi: 10.1111/cea.12343. Clin Exp Allergy. 2014. PMID: 24953633 No abstract available.
References
-
- Weber RW. Patterns of pollen cross-allergenicity. J Allergy Clin Immunol. 2003;112:229–39. - PubMed
-
- Weber RW. Cross-reactivity of pollen allergens. Curr Allergy Asthma Rep. 2004;4:401–8. - PubMed
-
- White JF, Bernstein DI. Key pollen allergens in North America. Ann Allergy Asthma Immunol. 2003;91:425–35. - PubMed
-
- Weber RW, Nelson HS. Pollen allergens and their interrelationships. Clin Rev Allergy. 1985;3:291–318. - PubMed
-
- Marcucci F, Sensi L, Di CG, Incorvaia C, Puccinelli P, Scurati S, Frati F. Which allergen extract for grass pollen immunotherapy? An in vitro study. Immunol Invest. 2010;39:635–44. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

