Lack of long-lasting hydrosalpinx in A/J mice correlates with rapid but transient chlamydial ascension and neutrophil recruitment in the oviduct following intravaginal inoculation with Chlamydia muridarum
- PMID: 24711570
- PMCID: PMC4097620
- DOI: 10.1128/IAI.00055-14
Lack of long-lasting hydrosalpinx in A/J mice correlates with rapid but transient chlamydial ascension and neutrophil recruitment in the oviduct following intravaginal inoculation with Chlamydia muridarum
Abstract
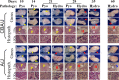
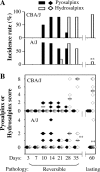
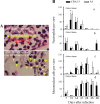
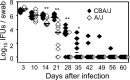
Lower genital tract infection with Chlamydia trachomatis and C. muridarum can induce long-lasting hydrosalpinx in the upper genital tract of women and female mice, respectively. However, A/J mice were highly resistant to induction of long-lasting hydrosalpinx by C. muridarum. We further compared host inflammatory responses and chlamydial infection courses between the hydrosalpinx-resistant A/J mice and CBA/J mice known to be susceptible to hydrosalpinx induction. Both mouse strains developed robust pyosalpinx during the acute phase followed by hydrosalpinx during the chronic phase. However, the hydrosalpinges disappeared in A/J mice by day 60 after infection, suggesting that some early hydrosalpinges are reversible. Although the overall inflammatory responses were indistinguishable between CBA/J and A/J mice, we found significantly more neutrophils in oviduct lumen of A/J mice on days 7 and 10, which correlated with a rapid but transient oviduct invasion by C. muridarum with a peak infection on day 7. In contrast, CBA/J mice developed a delayed and extensive oviduct infection. These comparisons have revealed an important role of the interactions of oviduct infection with inflammatory responses in chlamydial induction of long-lasting hydrosalpinx, suggesting that a rapid but transient invasion of oviduct by chlamydial organisms can prevent the development of the long-lasting hydrosalpinges.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures


Similar articles
-
Oviduct infection and hydrosalpinx in DBA1/j mice is induced by intracervical but not intravaginal inoculation with Chlamydia muridarum.PLoS One. 2013 Aug 5;8(8):e71649. doi: 10.1371/journal.pone.0071649. Print 2013. PLoS One. 2013. PMID: 23940777 Free PMC article.
-
Intrauterine infection with plasmid-free Chlamydia muridarum reveals a critical role of the plasmid in chlamydial ascension and establishes a model for evaluating plasmid-independent pathogenicity.Infect Immun. 2015 Jun;83(6):2583-92. doi: 10.1128/IAI.00353-15. Epub 2015 Apr 13. Infect Immun. 2015. PMID: 25870225 Free PMC article.
-
Chlamydial induction of hydrosalpinx in 11 strains of mice reveals multiple host mechanisms for preventing upper genital tract pathology.PLoS One. 2014 Apr 15;9(4):e95076. doi: 10.1371/journal.pone.0095076. eCollection 2014. PLoS One. 2014. PMID: 24736397 Free PMC article.
-
Chlamydia Spreading from the Genital Tract to the Gastrointestinal Tract - A Two-Hit Hypothesis.Trends Microbiol. 2018 Jul;26(7):611-623. doi: 10.1016/j.tim.2017.12.002. Epub 2017 Dec 27. Trends Microbiol. 2018. PMID: 29289422 Free PMC article. Review.
-
Regulation of chlamydial colonization by IFNγ delivered via distinct cells.Trends Microbiol. 2023 Mar;31(3):270-279. doi: 10.1016/j.tim.2022.09.002. Epub 2022 Sep 26. Trends Microbiol. 2023. PMID: 36175276 Free PMC article. Review.
Cited by
-
Chlamydia transmitting from the genital to gastrointestinal tract and inducing tubal disease: Double attack pattern.Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2022 Sep 28;47(9):1275-1280. doi: 10.11817/j.issn.1672-7347.2022.220023. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2022. PMID: 36411712 Free PMC article. Chinese, English.
-
In Vivo and Ex Vivo Imaging Reveals a Long-Lasting Chlamydial Infection in the Mouse Gastrointestinal Tract following Genital Tract Inoculation.Infect Immun. 2015 Sep;83(9):3568-77. doi: 10.1128/IAI.00673-15. Epub 2015 Jun 22. Infect Immun. 2015. PMID: 26099591 Free PMC article.
-
Effects of Immunomodulatory Drug Fingolimod (FTY720) on Chlamydia Dissemination and Pathogenesis.Infect Immun. 2020 Oct 19;88(11):e00281-20. doi: 10.1128/IAI.00281-20. Print 2020 Oct 19. Infect Immun. 2020. PMID: 32868341 Free PMC article.
-
Neutrophils Are Central to Antibody-Mediated Protection against Genital Chlamydia.Infect Immun. 2017 Sep 20;85(10):e00409-17. doi: 10.1128/IAI.00409-17. Print 2017 Oct. Infect Immun. 2017. PMID: 28739831 Free PMC article.
-
The cryptic plasmid is more important for Chlamydia muridarum to colonize the mouse gastrointestinal tract than to infect the genital tract.PLoS One. 2017 May 25;12(5):e0177691. doi: 10.1371/journal.pone.0177691. eCollection 2017. PLoS One. 2017. PMID: 28542376 Free PMC article.
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

